Advertisements
Advertisements
Question
Write structural formula for pentane-1,4-diol.
Advertisements
Solution
\[\begin{array}{cc}
\ce{H3C - HC - H2C - H2C - CH2 - OH}\\
|\phantom{......................}\\
\ce{\underset{Pentane-1,4-diol}{OH}\phantom{....................}}\\
\end{array}\]
RELATED QUESTIONS
What is metamerism?
Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\phantom{.....................................}\ce{CH2OH}\\
\phantom{............................}|\\
\ce{CH3 - CH - CH2 - CH - CH - CH3}\\
|\phantom{....................}|\phantom{............}\\
\ce{CH3}\phantom{..............}\ce{OH}\phantom{.........}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{H3C - CH - CH2 - CH - CH - CH2 - CH3}\\
|\phantom{....................}|\phantom{.........}|\phantom{.............}\\
\ce{OH}\phantom{................}\ce{OH}\phantom{...}\ce{C{_2}H5}\phantom{.........}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH3}\\
|\phantom{.........}|\phantom{...}\\
\ce{OH}\phantom{....}\ce{OH}\\
\end{array}\]
Write the IUPAC name of the following compound:
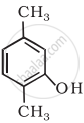
Write the IUPAC name of the following compound:
C6H5 – O – C7H15(n−)
Give the IUPAC name of the following ether:
CH3OCH2CH2Cl
Give the IUPAC name of the following ether:
CH3CH2CH2OCH3
How is phenol converted into the following?
picric acid
How do you convert the Ethanal to Propanone
In the dehydration of alcohols to alkenes by heating with concentrated sulphuric acid, the initiation step is:
(1) formation of carbonation
(2) formation of an ester
(3) protonation of the alcohol molecule
(4) elimination of water
Write structural formulae for 3-Methoxyhexane
Write IUPAC names of the following

Write IUPAC names of the following

Glycerol is ____________.
The product of acid catalysed hydration of 2-phenylpropene is:
Which of the following gives a positive iodoform test?
IUPAC name of the compound is:
\[\begin{array}{cc}
\ce{CH3-CH-OCH3}\\
|\phantom{....}\\
\ce{CH3}\phantom{..}
\end{array}\]
Arrange the following compounds in decreasing order of acidity.
\[\ce{H2O, ROH, HC ≡ CH}\]
Explain why p-nitrophenol is more acidic than phenol.
Assertion: Like bromination of benzene, bromination of phenol is also carried out in the presence of Lewis acid.
Reason: Lewis acid polarises the bromine molecule.
How can phenol be converted to aspirin?
Write the IUPAC name of the following compound.

Write a chemical reaction for the following conversion:
Acetic acid into ethyl alcohol.
Write structural formulae for:
p-Nitrophenol
Write structural formulae for:
Salicylic acid
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{...}OH\phantom{...}CH3}\\
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
