Advertisements
Advertisements
प्रश्न
Which of the following compounds is aromatic alcohol?
| (A) |  |
| (B) | 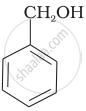 |
| (C) |  |
| (D) |  |
पर्याय
A, B, C, D
A, D
B, C
A
MCQ
Advertisements
उत्तर
B, C
Explanation:
Compound (A) i.e., phenol and compound (D) i.e., a derivative of phenol cannot be considered as aromatic alcohol. As phenol is also known as, carbolic acid cannot be considered as aromatic alcohol.
Compound (B) and (C), –OH group is bonded to sp3 hybridised carbon which in turn is bonded to benzene ring.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
