Advertisements
Advertisements
प्रश्न
How is phenol converted into the following?
Benzene
Convert the following:
Phenol into benzene.
Carry out the following conversion:
Phenol to benzene.
Write a chemical equation to convert the following:
Phenol to benzene.
Advertisements
उत्तर
When phenol is heated with zinc dust, it gets converted to benzene.

APPEARS IN
संबंधित प्रश्न
Explain metamerism with suitable examples of ethers
Write the IUPAC name of the given compound:

Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\phantom{..................}\ce{CH2OH}\\
\phantom{.........}|\\
\ce{CH3 - CH2 - CH - CH - CH - CH3}\\
\phantom{.........}|\phantom{...................}|\\
\phantom{.............}\ce{CH2Cl}\phantom{..........}\ce{CH3}\phantom{}
\end{array}\]
Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\ce{H2C = CH - CH - CH2 - CH2 - CH3}\\
|\phantom{...............}\\
\ce{OH}\phantom{............}
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{H3C - CH - CH2 - CH - CH - CH2 - CH3}\\
|\phantom{....................}|\phantom{.........}|\phantom{.............}\\
\ce{OH}\phantom{................}\ce{OH}\phantom{...}\ce{C{_2}H5}\phantom{.........}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH3}\\
|\phantom{.........}|\phantom{...}\\
\ce{OH}\phantom{....}\ce{OH}\\
\end{array}\]
Give the IUPAC name of the following ether:
\[\begin{array}{cc}
\ce{C2H5OCH2 - CH - CH3}\\
\phantom{.........}|\\
\phantom{.............}\ce{CH3}
\end{array}\]
Give the IUPAC name of the following ether:
CH3CH2CH2OCH3
What is the action of hot HI on it?
How is phenol converted into the following?
picric acid
Write the structures of the products when Butan-2-ol reacts with CrO3
Write the IUPAC name of the following :

Resorcinol on distillation with zinc dust gives _________.
Write structural formula for pentane-1,4-diol.
Write structural formulae for Cyclohex-2-en-1-ol.
Write IUPAC name of the following
\[\begin{array}{cc}\ce{CH3-CH-CH-CH2-OH}\\|\phantom{.....}|\phantom{.......}\\\ce{OH}\phantom{..}\ce{CH3}\phantom{.....}\end{array}\]
Write IUPAC names of the following

Give IUPAC names of the following compound:

C6H5OCH2CH3 is called:
Cresol has ____________.
Which of the following compounds is oxidised to prepare methyl ethyl ketone?
HBr reacts fastest with ____________.
Ethylene reacts with Baeyer’s reagent to give ______.
The IUPAC name of the ether CH2 = CH–CH2OCH3 is:
\[\ce{Phenol ->[Zn, dust] 'X' ->[CH3Cl][Anhy. AlCl3] 'Y' ->[Alkaline][KMnO4] 'Z'}\]
The product ‘Z’ is:
The correct acidic strength order of the following is:

(I)
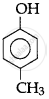
(II)

(III)
IUPAC name of the compound \[\begin{array}{cc}
\ce{CH3 - CH - OCH3}\\
\phantom{}|\phantom{....}\\
\phantom{}\ce{CH3}\phantom{..}
\end{array}\] is ______.
Give IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH - CH2 - CH2 - CH - CH3}\phantom{.}\\
\phantom{.........}|\phantom{...................}|\phantom{...........}\\
\phantom{..}\ce{Cl}\phantom{.................}\ce{OH}\phantom{..}
\end{array}\]
What happens when benzene diazonium chloride is heated with water?
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Assertion: IUPAC name of the compound
\[\begin{array}{cc}
\ce{CH3 - CH - O - CH2 - CH2 - CH3}\\
|\phantom{....................}\\
\ce{CH3}\phantom{.................}
\end{array}\] is 2-Ethoxy-2-methylethane.
Reason: In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by —OR or —OAr group [where R = alkyl group and Ar = aryl group]
Assertion: Phenol forms 2, 4, 6 – tribromophenol on treatment with \[\ce{Br2}\] in carbon disulphide at 273 K.
Reason: Bromine polarises in carbon disulphide.
Convert the following:
Ethyl alcohol into ethyl acetate.
Draw structure of the following compound.
2. 5-Diethylphenol
Write structural formulae for:
p-Nitrophenol
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{..............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{.....}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{.}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{...}OH\phantom{...}CH3}\\
\end{array}\]
