Advertisements
Advertisements
प्रश्न
Resorcinol on distillation with zinc dust gives _________.
पर्याय
Cyclohexane
Benzene
Toluene
Benzene-1, 3-diol
Advertisements
उत्तर
Resorcinol on distillation with zinc dust gives benzene.
Explanation:
Distillation of phenol with zinc dust gives benzene.
\[\ce{\underset{Phenol}{C6H5 - OH} + Zn ->[distillation] \underset{Benzene}{C6H6} + ZnO}\]
APPEARS IN
संबंधित प्रश्न
Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\phantom{.....................................}\ce{CH2OH}\\
\phantom{............................}|\\
\ce{CH3 - CH - CH2 - CH - CH - CH3}\\
|\phantom{....................}|\phantom{............}\\
\ce{CH3}\phantom{..............}\ce{OH}\phantom{.........}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{HO - CH2 - CH - CH2 - OH}\\
|\phantom{...}\\
\ce{OH}\phantom{.}\\
\end{array}\]
Write the IUPAC name of the following compound:

Write the IUPAC name of the following compound:
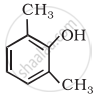
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - O - CH2 - CH - CH3}\\
\phantom{................}|\\
\phantom{....................}\ce{CH3}
\end{array}\]
Write the IUPAC name of the following compound:
C6H5 – O – C2H5
Write structures of the compounds whose IUPAC names are as follows:
3-Chloromethylpentan-1-ol.
Natalite is a mixture of
(a) diethyl ether and methanol
(b) diethyl ether and ethanol
(c) dimethyl ether and methanol
(d) dimethyl ether and ethanol
Propanoic acid to ethylamine.
Write IUPAC names of the following

In a carbinol system of nomenclature tert.butyl alcohol is named as _______________
Isopropyl alcohol on oxidation forms:
Give IUPAC names of the following compound:
\[\begin{array}{cc}
\phantom{..}\ce{H}\phantom{...}\ce{CH3}\phantom{.}\ce{H}\phantom{..}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - H}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\phantom{.}\ce{H}\phantom{...}\ce{OH}\phantom{.}\ce{H}\phantom{.}\\
\end{array}\]
One of the following is not a dihydroxy derivative of benzene.
Cresol has ____________.
The heating of phenyl methyl ether with HI produces:
Give IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH - CH2 - CH2 - CH - CH3}\phantom{.}\\
\phantom{.........}|\phantom{...................}|\phantom{...........}\\
\phantom{..}\ce{Cl}\phantom{.................}\ce{OH}\phantom{..}
\end{array}\]
Assertion: Addition reaction of water to but-1-ene in acidic medium yields butan-1-ol.
Reason: Addition of water in acidic medium proceeds through the formation of primary carbocation.
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Assertion: Like bromination of benzene, bromination of phenol is also carried out in the presence of Lewis acid.
Reason: Lewis acid polarises the bromine molecule.
Explain why Lewis acid is not required in bromination of phenol?
How can phenol be converted to aspirin?
Draw structure of the following compound.
2-Methoxypropane
Write structural formulae for:
Salicylic acid
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write the IUPAC name.
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C -CH3}\\
\phantom{.}|\phantom{......}|\phantom{......}|\\
\phantom{....}\ce{CH3\phantom{...}\ce{OH}\phantom{...}\ce{CH3}}\
\end{array}\]
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{...}OH\phantom{...}CH3}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
