Advertisements
Advertisements
प्रश्न
Write structures of the compounds whose IUPAC names are as follows:
3-Chloromethylpentan-1-ol.
Advertisements
उत्तर
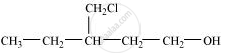
संबंधित प्रश्न
Write the IUPAC name of the following compound:
C6H5 – O – C7H15(n−)
Natalite is a mixture of
(a) diethyl ether and methanol
(b) diethyl ether and ethanol
(c) dimethyl ether and methanol
(d) dimethyl ether and ethanol
Write the structure and IUPAC name of 'methyl-n-propyl ether'.
Write the structures of the products when Butan-2-ol reacts with SOCl2
How do you convert the Ethanal to Propanone
What.will be the product fonned when chlorobenzene is heated with sodium metal in the presence of dry ether?
Write structural formulae for Cyclohex-2-en-1-ol.
Write IUPAC names of the following

Give IUPAC names of the following compound:

An example of a compound with functional group – O – is ____________.
Butane-2-ol is ____________.
Which of the following compounds is oxidised to prepare methyl ethyl ketone?
n-Propyl alcohol and isopropyl alcohol can be chemically distinguished by which reagent?
Ethylene reacts with Baeyer’s reagent to give ______.
The major product formed by the reaction:
\[\begin{array}{cc}
\ce{CH3CH-CH2Br ->[CH3O^-][CH3OH] is}\\
|\phantom{................}\\
\ce{CH3}\phantom{.............}
\end{array}\]
Write steps to carry out the conversion of phenol to aspirin.
Explain why p-nitrophenol is more acidic than phenol.
Convert the following:
Ethyl alcohol into ethyl acetate.
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{...}OH\phantom{...}CH3}\\
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
