Advertisements
Advertisements
प्रश्न
Write IUPAC name of the following compound:

Advertisements
उत्तर
2-Methylphenol
APPEARS IN
संबंधित प्रश्न
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{H3C - CH - CH2 - CH - CH - CH2 - CH3}\\
\phantom{}|\phantom{.............}|\phantom{......}|\phantom{.........}\\
\phantom{}\ce{OH}\phantom{..........}\ce{OH}\phantom{...}\ce{C2H5}\phantom{......}
\end{array}\]
Give IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
Give IUPAC name of the following ether:
How is phenol converted into the following?
Benzene
Write the IUPAC name of the following :

How do you convert the Ethanal to Propanone
Resorcinol on distillation with zinc dust gives _________.
Write structural formulae for 3-Methoxyhexane
Give IUPAC names of the following compound:

The compound HOCH2 – CH2OH is __________.
Butane-2-ol is ____________.
Which of the following compounds is oxidised to prepare methyl ethyl ketone?
Ethylene reacts with Baeyer’s reagent to give ______.
\[\ce{Phenol ->[Zn, dust] 'X' ->[CH3Cl][Anhy. AlCl3] 'Y' ->[Alkaline][KMnO4] 'Z'}\]
The product ‘Z’ is:
What happens when benzene diazonium chloride is heated with water?
Match the structures of the compounds given in Column I with the name of the compounds given in Column II.
| Column I | Column II | |
| (i) |  |
(a) Hydroquinone |
| (ii) |  |
(b) Phenetole |
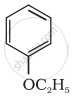
| (iii) | 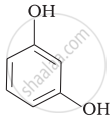 |
(c) Catechol |
| (iv) |  |
(d) o-Cresol |
| (v) |  |
(e) guinone |
| (vi) | 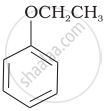 |
(f) Resorcinol |
| (g) Anisole |
Assertion: Addition reaction of water to but-1-ene in acidic medium yields butan-1-ol.
Reason: Addition of water in acidic medium proceeds through the formation of primary carbocation.
The IUPAC name of 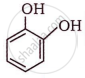 is ______.
is ______.
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
