Advertisements
Advertisements
प्रश्न
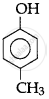
Write IUPAC name of the following compound:

Advertisements
उत्तर
2-Methylphenol
APPEARS IN
संबंधित प्रश्न
What is metamerism?
Name the following compound according to IUPAC system.

Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{HO - CH2 - CH - CH2 - OH}\\
|\phantom{..}\\
\ce{OH}
\end{array}\]
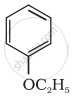
Write IUPAC name of the following compound:
C6H5 – O – C2H5
Give IUPAC name of the following ether:
CH3OCH2CH2Cl
Give IUPAC name of the following ether:
Write the structure and IUPAC name of 'methyl-n-propyl ether'.
How is phenol converted into the following?
picric acid
In the dehydration of alcohols to alkenes by heating with concentrated sulphuric acid, the initiation step is:
(1) formation of carbonation
(2) formation of an ester
(3) protonation of the alcohol molecule
(4) elimination of water
Write structural formula for pentane-1,4-diol.
An example of a compound with functional group – O – is ____________.
When ethyl alcohol reacts with acetic acid, the products formed are:
\[\ce{Phenol ->[Zn, dust] 'X' ->[CH3Cl][Anhy. AlCl3] 'Y' ->[Alkaline][KMnO4] 'Z'}\]
The product ‘Z’ is:
Among the following sets of reactants which one produces anisole?
The correct acidic strength order of the following is:

(I)
(II)

(III)
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Write complete reaction for the bromination of phenol in aqueous and non-aqueous medium.
Write structural formulae for:
Salicylic acid
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{..............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{.....}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{.}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write the IUPAC name.
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C -CH3}\\
\phantom{.}|\phantom{......}|\phantom{......}|\\
\phantom{....}\ce{CH3\phantom{...}\ce{OH}\phantom{...}\ce{CH3}}\
\end{array}\]
