Advertisements
Advertisements
Question
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Options
Assertion and reason both are correct and reason is correct explanation of assertion.
Assertion and reason both are wrong statements.
Assertion is correct statement but reason is wrong statement.
Assertion is wrong statement but reason is correct statement.
Both assertion and reason are correct statements but reason is not correct explanation of assertion.
Advertisements
Solution
Assertion and reason both are correct and reason is correct explanation of assertion.
Explanation:
P-nitro phenol is more acidic as nitro group helps in the stabilization of the phenoxide ion by dispersal of negative charge due to resonance.
APPEARS IN
RELATED QUESTIONS
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH3}\\
|\phantom{.........}|\phantom{...}\\
\ce{OH}\phantom{....}\ce{OH}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{HO - CH2 - CH - CH2 - OH}\\
|\phantom{...}\\
\ce{OH}\phantom{.}\\
\end{array}\]
Write IUPAC name of the following compound:

Give the IUPAC name of the following ether:
CH3OCH2CH2Cl
Give the IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
Give the IUPAC name of the following ether:

In the dehydration of alcohols to alkenes by heating with concentrated sulphuric acid, the initiation step is:
(1) formation of carbonation
(2) formation of an ester
(3) protonation of the alcohol molecule
(4) elimination of water
What.will be the product fonned when chlorobenzene is heated with sodium metal in the presence of dry ether?
Write IUPAC name of the following

Butane-2-ol is ____________.
When ethyl alcohol reacts with acetic acid, the products formed are:
Among the following sets of reactants which one produces anisole?
Write the IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH2 - C = C - OH}\\
\phantom{........}|\phantom{....}|\phantom{}\\
\phantom{..............}\ce{CH3 CH2OH}
\end{array}\]
Explain why p-nitrophenol is more acidic than phenol.
Assertion: IUPAC name of the compound
\[\begin{array}{cc}
\ce{CH3 - CH - O - CH2 - CH2 - CH3}\\
|\phantom{....................}\\
\ce{CH3}\phantom{.................}
\end{array}\] is 2-Ethoxy-2-methylethane.
Reason: In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by —OR or —OAr group [where R = alkyl group and Ar = aryl group]
Write a chemical reaction for the following conversion:
Acetic acid into ethyl alcohol.
Give the structures of Thiosulphuric acid and Peroxy monosulphuric acid.
Write structural formulae for:
p-Nitrophenol
The IUPAC name of 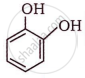 is ______.
is ______.
