Advertisements
Advertisements
प्रश्न
Assertion: Phenols give o- and p-nitrophenol on nitration with conc. \[\ce{HNO3}\] and \[\ce{H2SO4}\] mixture.
Reason: –OH group in phenol is o–, p– directing.
पर्याय
Assertion and reason both are correct and reason is correct explanation of assertion.
Assertion and reason both are wrong statements.
Assertion is correct statement but reason is wrong statement.
Assertion is wrong statement but reason is correct statement.
Both assertion and reason are correct statements but reason is not correct explanation of assertion.
Advertisements
उत्तर
Assertion is wrong statement but reason is correct statement.
Explanation:
Phenols give o, p-nitrophenol on nitration with dil. \[\ce{HNO3}\] and with cone.
• \[\ce{HNO3}\], 2, 4, 6-trinitrophenol is formed.
APPEARS IN
संबंधित प्रश्न
Write the IUPAC name of the given compound:

Write IUPAC name of the following compound:
C6H5 – O – C7H15(n−)
Give IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
Give IUPAC name of the following ether:
CH3CH2CH2OCH3
How is phenol converted into the following?
Benzene
How is phenol converted into the following?
benzoquinone
Write the structures of the products when Butan-2-ol reacts with CrO3
Propanoic acid to ethylamine.
Write structural formula for pentane-1,4-diol.
Write IUPAC name of the following

Write IUPAC names of the following

3-methylphenol is called ____________.
One of the following is not a dihydroxy derivative of benzene.
The compound HOCH2 – CH2OH is __________.
Butane-2-ol is ____________.
n-Propyl alcohol and isopropyl alcohol can be chemically distinguished by which reagent?
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Explain why Lewis acid is not required in bromination of phenol?
Give the structures of Thiosulphuric acid and Peroxy monosulphuric acid.
The IUPAC name of 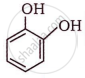 is ______.
is ______.
