Advertisements
Advertisements
प्रश्न
Match the starting materials given in Column I with the products formed by these (Column II) in the reaction with HI.
| Column I | Column II | ||
| (i) | CH3—O—CH3 | (a) |  |
| (ii) | \[\begin{array}{cc} \ce{CH3}\phantom{..................}\\ \backslash\phantom{.............}\\ \ce{CH-O-CH3}\\ /\phantom{..............}\\ \ce{CH3}\phantom{..................} \end{array}\] |
(b) | \[\begin{array}{cc} \ce{CH3}\phantom{....}\\ |\phantom{.......}\\ \ce{CH3-C-I + CH3OH}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
| (iii) | \[\begin{array}{cc} \ce{CH3}\phantom{.}\\ |\phantom{....}\\ \ce{H3C-C-O-CH3}\\ |\phantom{....}\\ \ce{CH3}\phantom{..} \end{array}\] |
(c) | 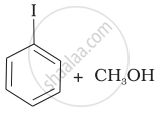 |
| (iv) |  |
(d) | CH3—OH + CH3—I |
| (e) | \[\begin{array}{cc} \ce{CH3}\phantom{.....................}\\ \backslash\phantom{.................}\\ \ce{CH-OH + CH3I}\\ /\phantom{.................}\\ \ce{CH3}\phantom{.....................} \end{array}\] |
||
| (f) | \[\begin{array}{cc} \ce{CH3}\phantom{.....................}\\ \backslash\phantom{.................}\\ \ce{CH-I + CH3OH}\\ /\phantom{.................}\\ \ce{CH3}\phantom{.....................} \end{array}\] |
||
| (g) | \[\begin{array}{cc} \ce{CH3}\phantom{....}\\ |\phantom{.......}\\ \ce{CH3-C-OH + CH3I}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
Advertisements
उत्तर
| Column I | Column II | ||
| (i) | CH3—O—CH3 | (d) | CH3—OH + CH3—I |
| (ii) | \[\begin{array}{cc} \ce{CH3}\phantom{..................}\\ \backslash\phantom{.............}\\ \ce{CH-O-CH3}\\ /\phantom{..............}\\ \ce{CH3}\phantom{..................} \end{array}\] |
(e) | \[\begin{array}{cc} \ce{CH3}\phantom{.....................}\\ \backslash\phantom{.................}\\ \ce{CH-OH + CH3I}\\ /\phantom{.................}\\ \ce{CH3}\phantom{.....................} \end{array}\] |
| (iii) | \[\begin{array}{cc} \ce{CH3}\phantom{.}\\ |\phantom{....}\\ \ce{H3C-C-O-CH3}\\ |\phantom{....}\\ \ce{CH3}\phantom{..} \end{array}\] |
(b) | \[\begin{array}{cc} \ce{CH3}\phantom{....}\\ |\phantom{.......}\\ \ce{CH3-C-I + CH3OH}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
| (iv) |  |
(a) |  |
Explanation:
(i) \[\ce{CH3 - O - CH3}\] is a symmetrical ether so the products are \[\ce{CH3I}\] and \[\ce{CH2OH}\].
(ii) In \[\ce{(CH3)2CH – O – CH3}\] unsymmetrical ether, one alkyl group is primary while another is secondary. So, it follows SN2 mechanism.
(iii) In this case, one of the alkyl group is tertiary and the other is primary. It follows SN1 mechanism and halide ion attacks the tertiary alkyl group and the products are \[\ce{(CH3)3 C-I}\] and \[\ce{CH3OH}\].
(iv) Here, the unsymmetrical ether is alkyl aryl ether. In this ether \[\ce{O - CH3}\] bond is weaker than \[\ce{O - C6H5}\] bond which has partial double bond character due to resonance. So, the halide ion attacks on alkyl group and the products are \[\ce{C6H5 - OH}\] and \[\ce{CH3I}\].
APPEARS IN
संबंधित प्रश्न
Explain metamerism with suitable examples of ethers
Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\phantom{..................}\ce{CH2OH}\\
\phantom{.........}|\\
\ce{CH3 - CH2 - CH - CH - CH - CH3}\\
\phantom{.........}|\phantom{...................}|\\
\phantom{.............}\ce{CH2Cl}\phantom{..........}\ce{CH3}\phantom{}
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{HO - CH2 - CH - CH2 - OH}\\
|\phantom{...}\\
\ce{OH}\phantom{.}\\
\end{array}\]
Write the IUPAC name of the following compound:
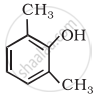
Write the IUPAC name of the following compound:

Write the IUPAC name of the following :

How do you convert the Ethanal to Propanone
In the dehydration of alcohols to alkenes by heating with concentrated sulphuric acid, the initiation step is:
(1) formation of carbonation
(2) formation of an ester
(3) protonation of the alcohol molecule
(4) elimination of water
In a carbinol system of nomenclature tert.butyl alcohol is named as _______________
The compound HOCH2 – CH2OH is __________.
Which of the following compounds is oxidised to prepare methyl ethyl ketone?
HBr reacts fastest with ____________.
Among the following sets of reactants which one produces anisole?
The correct acidic strength order of the following is:

(I)
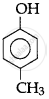
(II)

(III)
Arrange the following compounds in decreasing order of acidity.
\[\ce{H2O, ROH, HC ≡ CH}\]
Write complete reaction for the bromination of phenol in aqueous and non-aqueous medium.
Convert the following:
Ethyl alcohol into ethyl acetate.
How are the following conversions carried out?
Methyl magnesium bromide→2-Methylpropan-2-ol.
Write the IUPAC name.
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C -CH3}\\
\phantom{.}|\phantom{......}|\phantom{......}|\\
\phantom{....}\ce{CH3\phantom{...}\ce{OH}\phantom{...}\ce{CH3}}\
\end{array}\]
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{...}OH\phantom{...}CH3}\\
\end{array}\]
