Advertisements
Advertisements
प्रश्न
Which of the following is most acidic?
पर्याय
Benzyl alcohol
Cyclohexanol
Phenol
m-Chlorophenol
Advertisements
उत्तर
m-Chlorophenol
Explanation:
m-chlorophenol is most acidic. Alpha carbon of benzyl alcohol and cyclohexanol is sp3 hybridized. In m-chlorophenol, it is sp2 hybridized. In m-chlorophenol, electron-withdrawing group \[\ce{-Cl}\] is present at meta position.
APPEARS IN
संबंधित प्रश्न
Name the following compound according to IUPAC system.
\[\begin{array}{cc}
\phantom{............}\ce{CH2OH}\\
\phantom{......}|\\
\ce{CH3 - CH2 - CH - CH - CH - CH3}\\
\phantom{......}|\phantom{............}|\phantom{.}\\
\phantom{........}\ce{CH2Cl}\phantom{......}\ce{CH3}\phantom{}
\end{array}\]
Write IUPAC name of the following compound:

Write IUPAC name of the following compound:

Write IUPAC name of the following compound:
Write the structures of the products when Butan-2-ol reacts with CrO3
What.will be the product fonned when chlorobenzene is heated with sodium metal in the presence of dry ether?
3-methylphenol is called ____________.
The compound HOCH2 – CH2OH is __________.
Cresol has ____________.
Which of the following compounds is oxidised to prepare methyl ethyl ketone?
The IUPAC name of the ether CH2 = CH–CH2OCH3 is:
\[\ce{HC ≡ CH ->[HgSO4][H2SO4] ->[CH3MgBr][H2O] ->[PBr3]}\]
IUPAC name of m-cresol is ______.
Arrange the following compounds in decreasing order of acidity.
\[\ce{H2O, ROH, HC ≡ CH}\]
Assertion: Like bromination of benzene, bromination of phenol is also carried out in the presence of Lewis acid.
Reason: Lewis acid polarises the bromine molecule.
Write structural formulae for:
Salicylic acid
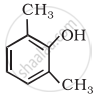
The IUPAC name of  is ______.
is ______.
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write the IUPAC name.
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C -CH3}\\
\phantom{.}|\phantom{......}|\phantom{......}|\\
\phantom{....}\ce{CH3\phantom{...}\ce{OH}\phantom{...}\ce{CH3}}\
\end{array}\]
