Advertisements
Advertisements
Question
Which of the following is most acidic?
Options
Benzyl alcohol
Cyclohexanol
Phenol
m-Chlorophenol
Advertisements
Solution
m-Chlorophenol
Explanation:
m-chlorophenol is most acidic. Alpha carbon of benzyl alcohol and cyclohexanol is sp3 hybridized. In m-chlorophenol, it is sp2 hybridized. In m-chlorophenol, electron-withdrawing group \[\ce{-Cl}\] is present at meta position.
APPEARS IN
RELATED QUESTIONS
Name the following compound according to IUPAC system.
\[\begin{array}{cc}
\phantom{........................}\ce{CH2OH}\\
\phantom{..................}|\\
\ce{CH3 - CH - CH2 - CH - CH - CH3}\\
\phantom{}|\phantom{.............}|\phantom{........}\\
\phantom{..}\ce{CH3}\phantom{..........}\ce{OH}\phantom{........}
\end{array}\]
Write IUPAC name of the following compound:
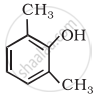
Write IUPAC name of the following compound:
C6H5 – O – C2H5
Give IUPAC name of the following ether:

Natalite is a mixture of
(a) diethyl ether and methanol
(b) diethyl ether and ethanol
(c) dimethyl ether and methanol
(d) dimethyl ether and ethanol
How is phenol converted into the following?
benzoquinone
Write the structures of the products when Butan-2-ol reacts with CrO3
In a carbinol system of nomenclature tert.butyl alcohol is named as _______________
Glycerol is ____________.
HBr reacts fastest with ____________.
Which of the following gives a positive iodoform test?
Which of the following compounds will react with sodium hydroxide solution in water?
What happens when benzene diazonium chloride is heated with water?
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Write complete reaction for the bromination of phenol in aqueous and non-aqueous medium.
Explain why Lewis acid is not required in bromination of phenol?
The IUPAC name of 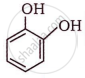 is ______.
is ______.
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{...}OH\phantom{...}CH3}\\
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
