Advertisements
Advertisements
प्रश्न
Which of the following is most acidic?
विकल्प
Benzyl alcohol
Cyclohexanol
Phenol
m-Chlorophenol
Advertisements
उत्तर
m-Chlorophenol
Explanation:
m-chlorophenol is most acidic. Alpha carbon of benzyl alcohol and cyclohexanol is sp3 hybridized. In m-chlorophenol, it is sp2 hybridized. In m-chlorophenol, electron-withdrawing group \[\ce{-Cl}\] is present at meta position.
APPEARS IN
संबंधित प्रश्न
Explain metamerism with suitable examples of ethers
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{HO - CH2 - CH - CH2 - OH}\\
|\phantom{..}\\
\ce{OH}
\end{array}\]
Write structures of the compounds whose IUPAC names are as follows:
3-Chloromethylpentan-1-ol.
Give IUPAC name of the following ether:

How is phenol converted into the following?
picric acid
Give reasons Fluoride ion has higher hydration enthalpy than chloride ion.
Glycerol is ____________.
Isopropyl alcohol on oxidation forms:
C6H5OCH2CH3 is called:
3-methylphenol is called ____________.
One of the following is not a dihydroxy derivative of benzene.
An example of a compound with functional group – O – is ____________.
Which of the following gives a positive iodoform test?
IUPAC name of m-cresol is ______.
IUPAC name of the compound \[\begin{array}{cc}
\ce{CH3 - CH - OCH3}\\
\phantom{}|\phantom{....}\\
\phantom{}\ce{CH3}\phantom{..}
\end{array}\] is ______.
Write the IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH2 - C = C - OH}\\
\phantom{........}|\phantom{....}|\phantom{}\\
\phantom{..............}\ce{CH3 CH2OH}
\end{array}\]
Write complete reaction for the bromination of phenol in aqueous and non-aqueous medium.
Explain why Lewis acid is not required in bromination of phenol?
Give the structures of Thiosulphuric acid and Peroxy monosulphuric acid.
The IUPAC name of 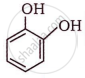 is ______.
is ______.
