Advertisements
Advertisements
प्रश्न
Write the mechanism of the following reaction :
\[\ce{C2H5OH->[H2SO4][443K]CH2=CH2 + H2O}\]
Advertisements
उत्तर
Mechanism: Dehydration of ethanol
Step 1: Formation of Protonated alcohol
Step 2: Formation of carbocation(slow step)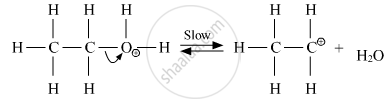
Step 3: Formation of ethane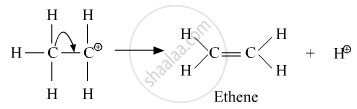
संबंधित प्रश्न
Among the following the one which reacts most readily with ethanol is:
If ethanol dissolves in water, then which of the following would be observed:
Write the IUPAC name of the following compound.
\[\begin{array}{cc}
\phantom{.}\ce{CH3 - CH - CH - CH - CH - CH3}\phantom{}\\
\phantom{......}|\phantom{......}|\phantom{......}|\phantom{.....}|\phantom{........}\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{C2H5}\phantom{.}\ce{OH}\phantom{...}\end{array}\]
What is denatured alcohol?
Suggest a reagent for conversion of ethanol to ethanal.
Suggest a reagent for conversion of ethanol to ethanoic acid.
Name the enzymes and write the reactions involved in the preparation of ethanol from sucrose by fermentation.
Liquor poisoning is due to
Alcoholic fermentation is brought about by the action of
3, 3-dimethyl-1-butene was subject to following reactions: