Advertisements
Advertisements
प्रश्न
Write the mechanism of the following reaction :
\[\ce{C2H5OH->[H2SO4][443K]CH2=CH2 + H2O}\]
Advertisements
उत्तर
Mechanism: Dehydration of ethanol
Step 1: Formation of Protonated alcohol
Step 2: Formation of carbocation(slow step)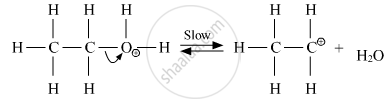
Step 3: Formation of ethane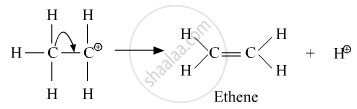
संबंधित प्रश्न
How do you convert the following:
Ethanol to Propan-2-ol
Give simple chemical test to distinguish between Ethanol and Phenol.
If ethanol dissolves in water, then which of the following would be observed:
Wood spirit is known as acetone:
Out of 2-chloroethanol and ethanol which is more acidic and why?
Suggest a reagent for conversion of ethanol to ethanoic acid.
Assertion: Ethanol is a weaker acid than phenol.
Reason: Sodium ethoxide may be prepared by the reaction of ethanol with aqueous \[\ce{NaOH}\].
Tonics in general contain
Alcoholic fermentation is brought about by the action of
Write chemical reactions of following reagents on methoxyethane:
dilute H2SO4
