Advertisements
Advertisements
प्रश्न
Match the items of column I with items of column II.
| Column I | Column II | |
| (i) | Methanol | (a) Conversion of phenol to o-hydroxysalicylic acid |
| (ii) | Kolbe’s reaction | (b) Ethyl alcohol |
| (iii) | Williamson’s synthesis | (c) Conversion of phenol to salicylaldehyde |
| (iv) | Conversion of 2° alcohol to ketone | (d) Wood spirit |
| (v) | Reimer-Tiemann reaction | (e) Heated copper at 573 K |
| (vi) | Fermentation | (f) Reaction of alkyl halide with sodium alkoxide |
Advertisements
उत्तर
| Column I | Column II | |
| (i) | Methanol | (d) Wood spirit |
| (ii) | Kolbe’s reaction | (a) Conversion of phenol to o-hydroxysalicylic acid |
| (iii) | Williamson’s synthesis | (f) Reaction of alkyl halide with sodium alkoxide |
| (iv) | Conversion of 2° alcohol to ketone | (e) Heated copper at 573 K |
| (v) | Reimer-Tiemann reaction | (c) Conversion of phenol to salicylaldehyde |
| (vi) | Fermentation | (b) Ethyl alcohol |
Explanation:
(i) Methanol is also known as ‘wood spirit’ as it was produced by the destructive distillation of wood.
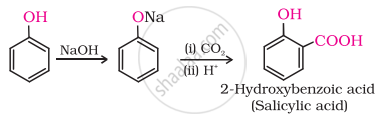
(ii) In Kolbe’s reaction, 2-hydroxybenzoic acid (salicylic acid) is prepared by the reaction of phenol with CO2 gas.
(iii) Williamson's synthesis is an important method for the preparation of either. In this method, an alkyl halide is allowed to react with sodium alkoxide.
\[\ce{R - X + R' - \overset{-}{\overset{\bullet\bullet}{\underset{\bullet\bullet}{O}}} \overset{+}{N}a -> R - \overset{\bullet\bullet}{\underset{\bullet\bullet}{O}} - R' + NaX}\]
(iv) When a 2° alcohol is allowed to pass overheated copper at 573 K, dehydrogenation takes place and an ketone is formed.
\[\begin{array}{cc}
\ce{R - CH - R' ->[Cu][573 K] R - C - R'}\\
|\phantom{...................}||\phantom{.}\\
\phantom{.}\ce{OH}\phantom{.................}\ce{O}\phantom{..}
\end{array}\]
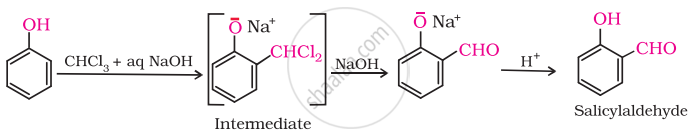
(v) On treating phenol with chloroform in the presence of \[\ce{NaOH}\], an aldehydic group is introduced at ortho position of benzene ring
(vi) Ethanol is prepared by the fermentation of sugars.
\[\ce{C12H22O11 + H2O ->[Invertase] C6H12O6 + C6H12O6}\]
\[\ce{C6H12O6 ->[Zymase] 2C2H5OH + 2CO2}\]
APPEARS IN
संबंधित प्रश्न
How do you convert the following:
Ethanol to Propan-2-ol
Give simple chemical test to distinguish between Ethanol and Phenol.
Methanol and ethanol are miscible in water due to ____________.
Wood spirit is known as acetone:
Phenol can be distinguished from ethanol by the reactions with:
(i) \[\ce{Br2/water}\]
(ii) \[\ce{Na}\]
(iii) Neutral \[\ce{FeCl3}\]
(iv) All the above
What is denatured alcohol?
Name the enzymes and write the reactions involved in the preparation of ethanol from sucrose by fermentation.
Dipole moment of phenol is smaller than that of methanol. Why?
Convert the following:
Propanenitrile to ethanol.
Which reagent can convert acetic acid into ethanol?
Tonics in general contain
Liquor poisoning is due to
Which of the following is known as wood spirit?
If the starting material is 1-methyl-1,2-epoxy cyclopentane, of absolute configuration, decide which one compound correctly represents the product of its reaction with sodium methoxide in methanol.
Convert the following Ethanal to ethanol.
How methanol is obtained from methanal.
Give IUPAC names of the following compounds:
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH - CH2-OH }\\
|\phantom{......}|\phantom{......}|\phantom{.......}\\
\ce{Cl}\phantom{....}\ce{CH3}\phantom{...}\ce{CH3}\phantom{.....}
\end{array}\]
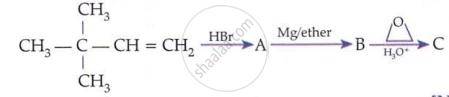
3, 3-dimethyl-1-butene was subject to following reactions: