Advertisements
Advertisements
Question
Match the items of column I with items of column II.
| Column I | Column II | |
| (i) | Methanol | (a) Conversion of phenol to o-hydroxysalicylic acid |
| (ii) | Kolbe’s reaction | (b) Ethyl alcohol |
| (iii) | Williamson’s synthesis | (c) Conversion of phenol to salicylaldehyde |
| (iv) | Conversion of 2° alcohol to ketone | (d) Wood spirit |
| (v) | Reimer-Tiemann reaction | (e) Heated copper at 573 K |
| (vi) | Fermentation | (f) Reaction of alkyl halide with sodium alkoxide |
Advertisements
Solution
| Column I | Column II | |
| (i) | Methanol | (d) Wood spirit |
| (ii) | Kolbe’s reaction | (a) Conversion of phenol to o-hydroxysalicylic acid |
| (iii) | Williamson’s synthesis | (f) Reaction of alkyl halide with sodium alkoxide |
| (iv) | Conversion of 2° alcohol to ketone | (e) Heated copper at 573 K |
| (v) | Reimer-Tiemann reaction | (c) Conversion of phenol to salicylaldehyde |
| (vi) | Fermentation | (b) Ethyl alcohol |
Explanation:
(i) Methanol is also known as ‘wood spirit’ as it was produced by the destructive distillation of wood.
(ii) In Kolbe’s reaction, 2-hydroxybenzoic acid (salicylic acid) is prepared by the reaction of phenol with CO2 gas.
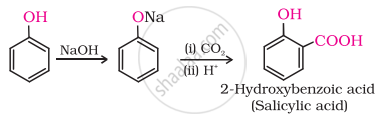
(iii) Williamson's synthesis is an important method for the preparation of either. In this method, an alkyl halide is allowed to react with sodium alkoxide.
\[\ce{R - X + R' - \overset{-}{\overset{\bullet\bullet}{\underset{\bullet\bullet}{O}}} \overset{+}{N}a -> R - \overset{\bullet\bullet}{\underset{\bullet\bullet}{O}} - R' + NaX}\]
(iv) When a 2° alcohol is allowed to pass overheated copper at 573 K, dehydrogenation takes place and an ketone is formed.
\[\begin{array}{cc}
\ce{R - CH - R' ->[Cu][573 K] R - C - R'}\\
|\phantom{...................}||\phantom{.}\\
\phantom{.}\ce{OH}\phantom{.................}\ce{O}\phantom{..}
\end{array}\]
(v) On treating phenol with chloroform in the presence of \[\ce{NaOH}\], an aldehydic group is introduced at ortho position of benzene ring
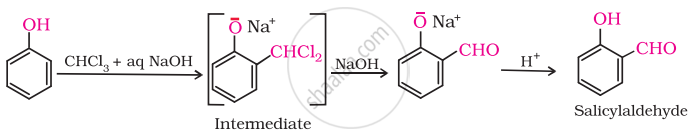
(vi) Ethanol is prepared by the fermentation of sugars.
\[\ce{C12H22O11 + H2O ->[Invertase] C6H12O6 + C6H12O6}\]
\[\ce{C6H12O6 ->[Zymase] 2C2H5OH + 2CO2}\]
APPEARS IN
RELATED QUESTIONS
Give simple chemical test to distinguish between Ethanol and Phenol.
Among the following the one which reacts most readily with ethanol is:
If ethanol dissolves in water, then which of the following would be observed:
Write the IUPAC name of the following compound.
\[\begin{array}{cc}
\phantom{.}\ce{CH3 - CH - CH - CH - CH - CH3}\phantom{}\\
\phantom{......}|\phantom{......}|\phantom{......}|\phantom{.....}|\phantom{........}\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{C2H5}\phantom{.}\ce{OH}\phantom{...}\end{array}\]
What is denatured alcohol?
Out of 2-chloroethanol and ethanol which is more acidic and why?
Suggest a reagent for conversion of ethanol to ethanal.
Suggest a reagent for conversion of ethanol to ethanoic acid.
Arrange the following compounds in increasing order of acidity and give a suitable explanation.
Phenol, o-nitrophenol, o-cresol
Name the enzymes and write the reactions involved in the preparation of ethanol from sucrose by fermentation.
The carbon-oxygen bond in phenol is slightly stronger than that in methanol. Why?
Which reagent can convert acetic acid into ethanol?
Alcoholic fermentation is brought about by the action of
Which of the following is known as wood spirit?
Write chemical reactions of following reagents on methoxyethane:
dilute H2SO4
If the starting material is 1-methyl-1,2-epoxy cyclopentane, of absolute configuration, decide which one compound correctly represents the product of its reaction with sodium methoxide in methanol.
Convert the following Ethanal to ethanol.
