Advertisements
Advertisements
Question
The carbon-oxygen bond in phenol is slightly stronger than that in methanol. Why?
Advertisements
Solution
This can be explained as under:
(i) In phenol, the conjugation of unshared electron pairs over oxygen with aromatic ring results in partial double bond character in C – O bond.
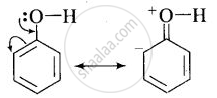
In methanol, no such conjugation (resonance) is possible.
(ii) In phenol, oxygen is attached to sp2 hybridised carbon while in methanol, oxygen attached to sp2 hybridised carbon. An sp2 hybridised carbon is more electronegative (because of greater 5-character) than sp3 hybridised carbon atom. Therefore, the bond between oxygen and sp2 hybridised carbon is more stable than the bond between oxygen and sp2, hybridised orbital.
APPEARS IN
RELATED QUESTIONS
How do you convert the following:
Ethanol to Propan-2-ol
Among the following the one which reacts most readily with ethanol is:
Methanol and ethanol are miscible in water due to ____________.
If ethanol dissolves in water, then which of the following would be observed:
Which of the following is correct?
Wood spirit is known as acetone:
Phenol is less acidic than ______.
Write the IUPAC name of the following compound.
\[\begin{array}{cc}
\phantom{.}\ce{CH3 - CH - CH - CH - CH - CH3}\phantom{}\\
\phantom{......}|\phantom{......}|\phantom{......}|\phantom{.....}|\phantom{........}\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{C2H5}\phantom{.}\ce{OH}\phantom{...}\end{array}\]
Suggest a reagent for conversion of ethanol to ethanal.
Dipole moment of phenol is smaller than that of methanol. Why?
Arrange water, ethanol and phenol in increasing order of acidity and give reason for your answer.
Match the items of column I with items of column II.
| Column I | Column II | |
| (i) | Methanol | (a) Conversion of phenol to o-hydroxysalicylic acid |
| (ii) | Kolbe’s reaction | (b) Ethyl alcohol |
| (iii) | Williamson’s synthesis | (c) Conversion of phenol to salicylaldehyde |
| (iv) | Conversion of 2° alcohol to ketone | (d) Wood spirit |
| (v) | Reimer-Tiemann reaction | (e) Heated copper at 573 K |
| (vi) | Fermentation | (f) Reaction of alkyl halide with sodium alkoxide |
Assertion: Ethanol is a weaker acid than phenol.
Reason: Sodium ethoxide may be prepared by the reaction of ethanol with aqueous \[\ce{NaOH}\].
Alcoholic fermentation is brought about by the action of
Which of the following is known as wood spirit?
Convert the following Ethanal to ethanol.
How methanol is obtained from methanal.
