Advertisements
Advertisements
प्रश्न
The carbon-oxygen bond in phenol is slightly stronger than that in methanol. Why?
Advertisements
उत्तर
This can be explained as under:
(i) In phenol, the conjugation of unshared electron pairs over oxygen with aromatic ring results in partial double bond character in C – O bond.
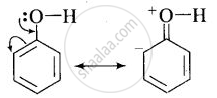
In methanol, no such conjugation (resonance) is possible.
(ii) In phenol, oxygen is attached to sp2 hybridised carbon while in methanol, oxygen attached to sp2 hybridised carbon. An sp2 hybridised carbon is more electronegative (because of greater 5-character) than sp3 hybridised carbon atom. Therefore, the bond between oxygen and sp2 hybridised carbon is more stable than the bond between oxygen and sp2, hybridised orbital.
APPEARS IN
संबंधित प्रश्न
How do you convert the following:
Ethanol to Propan-2-ol
Write the mechanism of the following reaction :
\[\ce{C2H5OH->[H2SO4][443K]CH2=CH2 + H2O}\]
Give simple chemical test to distinguish between Ethanol and Phenol.
Methanol and ethanol are miscible in water due to ____________.
Phenol is less acidic than ______.
What is denatured alcohol?
Suggest a reagent for conversion of ethanol to ethanoic acid.
Name the enzymes and write the reactions involved in the preparation of ethanol from sucrose by fermentation.
Dipole moment of phenol is smaller than that of methanol. Why?
Assertion: Ethanol is a weaker acid than phenol.
Reason: Sodium ethoxide may be prepared by the reaction of ethanol with aqueous \[\ce{NaOH}\].
Which reagent can convert acetic acid into ethanol?
Tonics in general contain
Liquor poisoning is due to
Convert the following Ethanal to ethanol.
How methanol is obtained from methanal.
Give IUPAC names of the following compounds:
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH - CH2-OH }\\
|\phantom{......}|\phantom{......}|\phantom{.......}\\
\ce{Cl}\phantom{....}\ce{CH3}\phantom{...}\ce{CH3}\phantom{.....}
\end{array}\]
3, 3-dimethyl-1-butene was subject to following reactions: