Advertisements
Advertisements
Question
Match the items of column I with items of column II.
| Column I | Column II | |
| (i) | Antifreeze used in car engine | (a) Neutral ferric chloride |
| (ii) | Solvent used in perfumes | (b) Glycerol |
| (iii) | Starting material for picric acid | (c) Methanol |
| (iv) | Wood spirit | (d) Phenol |
| (v) | Reagent used for detection of phenolic group | (e) Ethleneglycol |
| (vi) | By product of soap industry used in cosmetics | (f) Ethanol |
Advertisements
Solution
| Column I | Column II | |
| (i) | Antifreeze used in car engine | (e) Ethleneglycol |
| (ii) | Solvent used in perfumes | (f) Ethanol |
| (iii) | Starting material for picric acid | (d) Phenol |
| (iv) | Wood spirit | (c) Methanol |
| (v) | Reagent used for detection of phenolic group | (a) Neutral ferric chloride |
| (vi) | By product of soap industry used in cosmetics | (b) Glycerol |
Explanation:
(i) IUPAC name of ethylene glycol is ethane-1, 2-diol. It is primarily used as raw material in the manufacturing of polyester fibers and fabric industry. A small percentage of it is used in antifreeze formulations.
(ii) Ethanol is a good solvent for fatty and waxy substances. Fats and waxes provide odour to the perfumes. Apart from being a good solvent, it is less irritating to the skin. So, it is used in perfumes.
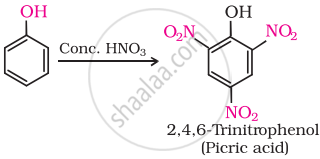
(iii) Phenol is converted into picric acid (2, 4, 6-trinitro-phenol) by the reaction of phenol with cone. \[\ce{HNO3}\].
(iv) Methanol, \[\ce{CH3OH}\] is also known as ‘wood spirit’ as it was produced by the destructive distillation of wood.
(v) Neutral ferric chloride give purple/red colour when treated with phenols. It is the reagent used for detection of phenolic group.
(vi) Soaps are prepared by the reactions of fatty acid with \[\ce{NaOH}\].
\[\begin{array}{cc}
\ce{O}\phantom{..........................................}\\
||\phantom{..........................................}\\
\ce{CH2 - O - C - OC17H35}\phantom{...........................}\phantom{.......}\ce{CH2OH}\phantom{.}\\
\phantom{.}
\phantom{.........}|\phantom{..........}\ce{O}\phantom{.............................................}|\phantom{.................}\\
||\phantom{...........................................}\\
\ce{CH - O - C - OC17H35 + 3NaOH -> \underset{(soap)}{\underset{Sodium stearate}{3C17H35COONa}} + CHOH}\phantom{..}\\
\phantom{..}|\phantom{.........................................................}|\phantom{.........}\\
\phantom{}\ce{CH2 - O - C -OC17H35}\phantom{..................................}\ce{\underset{Glycerol}{CH2OH}}\phantom{}\\
||\phantom{........................................}\\
\ce{O}\phantom{........................................}\\
\end{array}\]
The glycerol (propan-1, 2, 3-triol) is the by product of saop industry and used in cosmetics.
APPEARS IN
RELATED QUESTIONS
Give simple chemical test to distinguish between Ethanol and Phenol.
Among the following the one which reacts most readily with ethanol is:
Methanol and ethanol are miscible in water due to ____________.
If ethanol dissolves in water, then which of the following would be observed:
Wood spirit is known as acetone:
Suggest a reagent for conversion of ethanol to ethanoic acid.
Name the enzymes and write the reactions involved in the preparation of ethanol from sucrose by fermentation.
Dipole moment of phenol is smaller than that of methanol. Why?
The carbon-oxygen bond in phenol is slightly stronger than that in methanol. Why?
Arrange water, ethanol and phenol in increasing order of acidity and give reason for your answer.
Convert the following:
Propanenitrile to ethanol.
Liquor poisoning is due to
Which of the following is known as wood spirit?
Write chemical reactions of following reagents on methoxyethane:
dilute H2SO4
Convert the following Ethanal to ethanol.
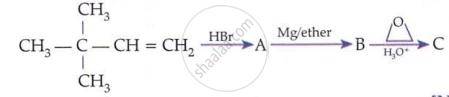
3, 3-dimethyl-1-butene was subject to following reactions: