Advertisements
Advertisements
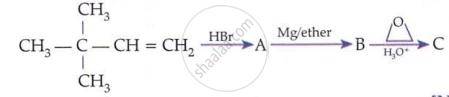
प्रश्न
Suggest a reagent for conversion of ethanol to ethanal.
Advertisements
उत्तर
\[\ce{CrO3}\], pyridine and HCl (Pyridinium chlorochromate, PCC) in \[\ce{CH2C12}\].
APPEARS IN
संबंधित प्रश्न
How do you convert the following:
Ethanol to Propan-2-ol
Write the mechanism of the following reaction :
\[\ce{C2H5OH->[H2SO4][443K]CH2=CH2 + H2O}\]
Give simple chemical test to distinguish between Ethanol and Phenol.
Among the following the one which reacts most readily with ethanol is:
If ethanol dissolves in water, then which of the following would be observed:
Phenol is less acidic than ______.
Phenol can be distinguished from ethanol by the reactions with:
(i) \[\ce{Br2/water}\]
(ii) \[\ce{Na}\]
(iii) Neutral \[\ce{FeCl3}\]
(iv) All the above
Write the IUPAC name of the following compound.
\[\begin{array}{cc}
\phantom{.}\ce{CH3 - CH - CH - CH - CH - CH3}\phantom{}\\
\phantom{......}|\phantom{......}|\phantom{......}|\phantom{.....}|\phantom{........}\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{C2H5}\phantom{.}\ce{OH}\phantom{...}\end{array}\]
Out of 2-chloroethanol and ethanol which is more acidic and why?
Suggest a reagent for conversion of ethanol to ethanoic acid.
Name the enzymes and write the reactions involved in the preparation of ethanol from sucrose by fermentation.
Match the items of column I with items of column II.
| Column I | Column II | |
| (i) | Antifreeze used in car engine | (a) Neutral ferric chloride |
| (ii) | Solvent used in perfumes | (b) Glycerol |
| (iii) | Starting material for picric acid | (c) Methanol |
| (iv) | Wood spirit | (d) Phenol |
| (v) | Reagent used for detection of phenolic group | (e) Ethleneglycol |
| (vi) | By product of soap industry used in cosmetics | (f) Ethanol |
Assertion: Ethanol is a weaker acid than phenol.
Reason: Sodium ethoxide may be prepared by the reaction of ethanol with aqueous \[\ce{NaOH}\].
Liquor poisoning is due to
Write chemical reactions of following reagents on methoxyethane:
dilute H2SO4
If the starting material is 1-methyl-1,2-epoxy cyclopentane, of absolute configuration, decide which one compound correctly represents the product of its reaction with sodium methoxide in methanol.
How methanol is obtained from methanal.
3, 3-dimethyl-1-butene was subject to following reactions: