Advertisements
Advertisements
प्रश्न
Write resonance structures of aniline. What is the action of benzene diazonium chloride on ethanol?
Advertisements
उत्तर
a. Resonance structure of aniline:
In aniline and other arylamines, the amino group is directly attached to benzene ring. Conjugation of lone pair of electrons stabilizes aniline. The lone pair is not easily available for protonation. Five resonance structures of aniline are as follows:

b. Action of benzene diazonium chloride on ethanol:
Reduction of benzene diazonium chloride in the presence of ethanol gives benzene. The ethanol gets oxidized to ethanal.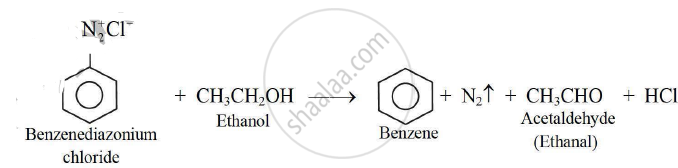
APPEARS IN
संबंधित प्रश्न
How do you convert the following:
Ethanol to Propan-2-ol
Write the mechanism of the following reaction :
\[\ce{C2H5OH->[H2SO4][443K]CH2=CH2 + H2O}\]
Give simple chemical test to distinguish between Ethanol and Phenol.
Among the following the one which reacts most readily with ethanol is:
Methanol and ethanol are miscible in water due to ____________.
If ethanol dissolves in water, then which of the following would be observed:
Which of the following is correct?
Phenol is less acidic than ______.
Phenol can be distinguished from ethanol by the reactions with:
(i) \[\ce{Br2/water}\]
(ii) \[\ce{Na}\]
(iii) Neutral \[\ce{FeCl3}\]
(iv) All the above
Write the IUPAC name of the following compound.
\[\begin{array}{cc}
\phantom{.}\ce{CH3 - CH - CH - CH - CH - CH3}\phantom{}\\
\phantom{......}|\phantom{......}|\phantom{......}|\phantom{.....}|\phantom{........}\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{C2H5}\phantom{.}\ce{OH}\phantom{...}\end{array}\]
Suggest a reagent for conversion of ethanol to ethanal.
Suggest a reagent for conversion of ethanol to ethanoic acid.
Name the enzymes and write the reactions involved in the preparation of ethanol from sucrose by fermentation.
Dipole moment of phenol is smaller than that of methanol. Why?
The carbon-oxygen bond in phenol is slightly stronger than that in methanol. Why?
Arrange water, ethanol and phenol in increasing order of acidity and give reason for your answer.
Match the items of column I with items of column II.
| Column I | Column II | |
| (i) | Antifreeze used in car engine | (a) Neutral ferric chloride |
| (ii) | Solvent used in perfumes | (b) Glycerol |
| (iii) | Starting material for picric acid | (c) Methanol |
| (iv) | Wood spirit | (d) Phenol |
| (v) | Reagent used for detection of phenolic group | (e) Ethleneglycol |
| (vi) | By product of soap industry used in cosmetics | (f) Ethanol |
Assertion: Ethanol is a weaker acid than phenol.
Reason: Sodium ethoxide may be prepared by the reaction of ethanol with aqueous \[\ce{NaOH}\].
Convert the following:
Propanenitrile to ethanol.
Tonics in general contain
Alcoholic fermentation is brought about by the action of
Which of the following is known as wood spirit?
Write chemical reactions of following reagents on methoxyethane:
dilute H2SO4
Give IUPAC names of the following compounds:
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH - CH2-OH }\\
|\phantom{......}|\phantom{......}|\phantom{.......}\\
\ce{Cl}\phantom{....}\ce{CH3}\phantom{...}\ce{CH3}\phantom{.....}
\end{array}\]
3, 3-dimethyl-1-butene was subject to following reactions: