Advertisements
Advertisements
Question
Explain why nucleophilic substitution reactions are not very common in phenols.
Advertisements
Solution
The C – O bond in phenols has some double-bond character due to resonance and hence cannot be easily cleaved by nucleophiles. So, nucleophilic substitution reactions are not very common in phenols and they give many electrophilic substitution reactions.
APPEARS IN
RELATED QUESTIONS
Write the mechanism of the following reaction :

Give the equation of the following reaction:
Oxidation of propan-1-ol with alkaline KMnO4 solution.
Give the equation of the following reaction:
Dilute HNO3 with phenol.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
When 3-methylbutan-2-ol is treated with HBr, the following reaction takes place:
\[\begin{array}{cc}
\phantom{.......................}\ce{Br}\\
\phantom{......................}|\\
\ce{CH3 - CH - CH - CH3 ->[HBr] CH3 - C - CH2 - CH3}\\
\phantom{.}|\phantom{......}|\phantom{......................}|\phantom{........}\\
\phantom{}\ce{CH3}\phantom{...}\ce{OH}\phantom{...................}\ce{CH3}\phantom{.....}
\end{array}\]
Give a mechanism for this reaction.
(Hint: The secondary carbocation formed in step II rearranges to a more
stable tertiary carbocation by a hydride ion shift from 3rd carbon atom.)
Lucas reagent is ____________.
In the reduction \[\ce{R - CHO + H2 -> RCH2OH}\] the catalyst used is:
By which of the following methods alcohol can be prepared in excellent yield?
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
Which one of the following on oxidation gives a ketone?
The compound which gives the most stable carbonium ion on dehydration is:
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
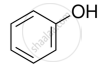
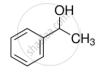
The process of converting alkyl halides into alcohols involves ______.
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) |  |
| (b) |  |
| (c) |  |
Why is the C – O – H bond angle in alcohols slightly less than the tetrahedral angle whereas the C – O – C bond angle in ether is slightly greater?
Which of the following alcohols will not undergo oxidation?
Write the mechanism of acid dehydration of ethanol to yield ethene.
