Advertisements
Advertisements
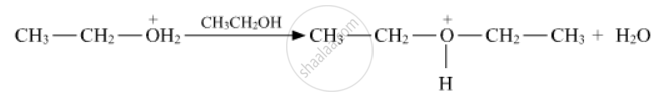
Question
Write the mechanism of the following reaction :

Advertisements
Solution 1
Primary alcohols such as ethanol react by SN2 mechanism with halogen acid such as HBr as follows:

Solution 2
This is a nucleophilic substitution reaction. It follows SN2 mechanism. The mechanism is as follows:
1) Protonation of hydroxyl group to make it a good leaving group.
2) Displacement of water molecule by bromide ion to form alkyl bromide.

APPEARS IN
RELATED QUESTIONS
Write the final product(s) in each of the following reactions:

Write the mechanism of the following reaction :

When 3-methylbutan-2-ol is treated with HBr, the following reaction takes place:
\[\begin{array}{cc}
\phantom{...................................}\ce{Br}\\
\phantom{..................................}|\\
\ce{CH3 - CH - CH - CH3 ->[HBr] CH3 - C - CH2 - CH3}\\
|\phantom{.........}|\phantom{...................................}|\phantom{...........}\\
\ce{CH3}\phantom{...}\ce{OH}\phantom{...............................}\ce{CH3}\phantom{.......}\\
\end{array}\]
Give a mechanism for this reaction.
(Hint: The secondary carbocation formed in step II rearranges to a more
stable tertiary carbocation by a hydride ion shift from 3rd carbon atom.)
Write the mechanism (using curved arrow notation) of the following reaction :
Explain why nucleophilic substitution reactions are not very common in phenols.
In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
Why is the C – O – H bond angle in alcohols slightly less than the tetrahedral angle whereas the C – O – C bond angle in ether is slightly greater?
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene
