Advertisements
Advertisements
Question
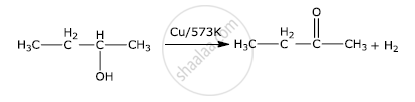
Write the final product(s) in each of the following reactions:

Advertisements
Solution
APPEARS IN
RELATED QUESTIONS
Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Give the equation of the following reaction:
Dilute HNO3 with phenol.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to aldehyde.
When 3-methylbutan-2-ol is treated with HBr, the following reaction takes place:
\[\begin{array}{cc}
\phantom{...................................}\ce{Br}\\
\phantom{..................................}|\\
\ce{CH3 - CH - CH - CH3 ->[HBr] CH3 - C - CH2 - CH3}\\
|\phantom{.........}|\phantom{...................................}|\phantom{...........}\\
\ce{CH3}\phantom{...}\ce{OH}\phantom{...............................}\ce{CH3}\phantom{.......}\\
\end{array}\]
Give a mechanism for this reaction.
(Hint: The secondary carbocation formed in step II rearranges to a more
stable tertiary carbocation by a hydride ion shift from 3rd carbon atom.)
The process of converting alkyl halides into alcohols involves ______.
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) |  |
| (b) |  |
| (c) |  |
What happens when (CH3)3 C – OH is heated with Cu/573 K?
Write the chemical equation in support of your answer.
Which of the following alcohols will not undergo oxidation?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
