Advertisements
Advertisements
Question
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to aldehyde.
Advertisements
Solution
Pyridinium chlorochromate (PCC), C5H5NH+Cr CrO3Cl− (in CH2Cl2) or pyridinium dichromate (PDC), \[\ce{(C5H5N+H)2Cr2O^-_7}\] (in CH2Cl2) is used in the oxidation of a primary alcohol to aldehyde.
APPEARS IN
RELATED QUESTIONS
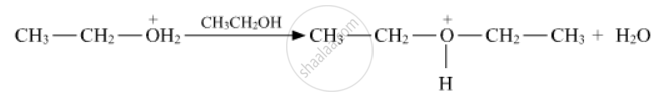
Write the mechanism (using curved arrow notation) of the following reaction :
Lucas reagent is ____________.
Lucas test is used for the detection of _____________.
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
Lucas test is done to differentiate between ____________.
Primary and secondary alcohols on the action of reduced copper give:
\[\ce{CH3CH2OH}\] can be converted into \[\ce{CH3CHO}\] by ______.
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
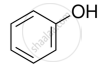
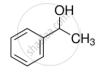
The process of converting alkyl halides into alcohols involves ______.
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) |  |
| (b) |  |
| (c) |  |
Alcohols react with active metals e.g. Na, K etc. to give corresponding alkoxides. Write down the decreasing order of reactivity of sodium metal towards primary, secondary and tertiary alcohols.
Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
What is Lucas reagent?
What happens when (CH3)3 C – OH is heated with Cu/573 K?
Write the chemical equation in support of your answer.
Which of the following observation is shown by 2-phenyl ethanol with Lucas Reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
