Advertisements
Advertisements
Question
Which of the following observation is shown by 2-phenyl ethanol with Lucas Reagent?
Options
Turbidity will be observed within five minutes.
No turbidity will be observed.
Turbidity will be observed immediately.
Turbidity will be observed at room temperature but will disappear after five minutes.
Advertisements
Solution
No turbidity will be observed.
Explanation:
Alcohols are soluble in Lucas reagent (conc. HCl and ZnCl2) while their halides are immiscible and produce turbidity in solution. In case of tertiary alcohols, turbidity is produced immediately as they form the halides easily. Primary alcohols do not produce turbidity at room temperature.
APPEARS IN
RELATED QUESTIONS
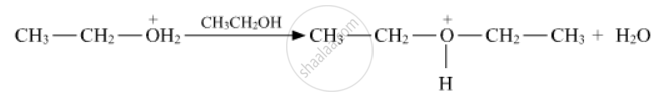
Write the mechanism of the following reaction:

Name the reagent used in the following reaction:
Oxidation of a primary alcohol to aldehyde.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
Write the mechanism (using curved arrow notation) of the following reaction :
The compound which reacts fastest with Lucas reagent at room temperature is:
Lucas test is done to differentiate between ____________.
Cyclohexene is best prepared from cyclohexanol by which of the following:
The correct geometry around oxygen in CH3OCH3 is
Which of the following alcohols will not undergo oxidation?
Write the mechanism of acid dehydration of ethanol to yield ethene.
