Advertisements
Advertisements
Question
Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
Advertisements
Solution
In phenol, oxygen atom is attached to sp2-hybridised carbon atom while in alcohol, it is attached to sp3-hybridised carbon atom. The bond formed between oxygen and sp2-hybridised carbon is more strongly held then that formed between oxygen and sp3-hybridised carbon.
APPEARS IN
RELATED QUESTIONS
Write the final product(s) in each of the following reactions:

Write the mechanism of the following reaction :

Give the equation of the following reaction:
Dilute HNO3 with phenol.
Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
Lucas reagent is ____________.
Lucas test is used for the detection of _____________.
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
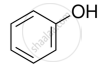
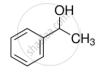
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) |  |
| (b) |  |
| (c) |  |
Suggest a reagent for the following conversion.

Why is the C – O – H bond angle in alcohols slightly less than the tetrahedral angle whereas the C – O – C bond angle in ether is slightly greater?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
