Advertisements
Advertisements
Question
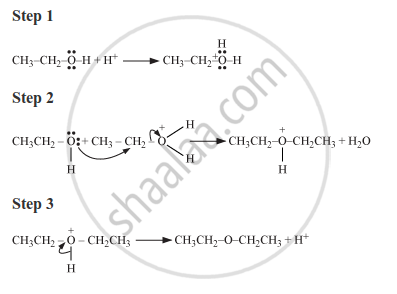
Write the mechanism of the following reaction :

Advertisements
Solution

The given reaction follows SN2 mechanism as shown below:
APPEARS IN
RELATED QUESTIONS
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Write the mechanism of the following reaction :

Lucas test is done to differentiate between ____________.
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
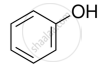
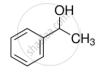
Suggest a reagent for the following conversion.

Explain why nucleophilic substitution reactions are not very common in phenols.
Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
What is Lucas reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
