Advertisements
Advertisements
प्रश्न
Write the mechanism of the following reaction :

Advertisements
उत्तर

The given reaction follows SN2 mechanism as shown below:
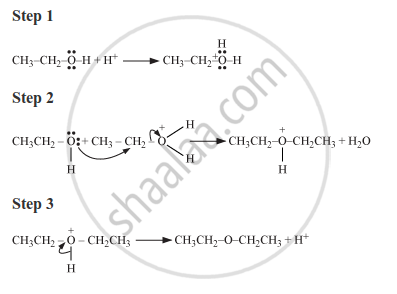
APPEARS IN
संबंधित प्रश्न
Write the mechanism of the following reaction:

Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
Primary and secondary alcohols on the action of reduced copper give:
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) |  |
| (b) |  |
| (c) |  |
Name the factors responsible for the solubility of alcohols in water.
Assertion: Bond angle in ethers is slightly less than the tetrahedral angle.
Reason: There is a repulsion between the two bulky (–R) groups.
Which of the following observation is shown by 2-phenyl ethanol with Lucas Reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
