Advertisements
Advertisements
प्रश्न
Name the factors responsible for the solubility of alcohols in water.
Advertisements
उत्तर
Alcohols are soluble in water because of hydrogen bonding with water molecules.
\[\begin{array}{cc}
\phantom{}\ce{R - O - H ⋅⋅⋅ O - H ⋅⋅⋅ O - H ⋅⋅⋅ }\phantom{}\\
\phantom{.......}|\phantom{..........}|\phantom{.}\\
\phantom{.......}\ce{H}\phantom{.........}\ce{R}\phantom{.}
\end{array}\]
The solubility decreases with increase in size of alkyl or aryl group.
APPEARS IN
संबंधित प्रश्न
Write the final product(s) in each of the following reactions:

Write the mechanism of the following reaction :

Show how will you synthesize pentan-1-ol using a suitable alkyl halide.
Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
When 3-methylbutan-2-ol is treated with HBr, the following reaction takes place:
\[\begin{array}{cc}
\phantom{...................................}\ce{Br}\\
\phantom{..................................}|\\
\ce{CH3 - CH - CH - CH3 ->[HBr] CH3 - C - CH2 - CH3}\\
|\phantom{.........}|\phantom{...................................}|\phantom{...........}\\
\ce{CH3}\phantom{...}\ce{OH}\phantom{...............................}\ce{CH3}\phantom{.......}\\
\end{array}\]
Give a mechanism for this reaction.
(Hint: The secondary carbocation formed in step II rearranges to a more
stable tertiary carbocation by a hydride ion shift from 3rd carbon atom.)
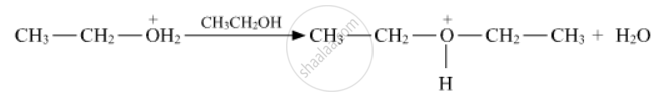
Write the mechanism (using curved arrow notation) of the following reaction :
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
Which of the following is not true in case of reaction with heated copper at 300°C?
Cyclohexene is best prepared from cyclohexanol by which of the following:
Suggest a reagent for the following conversion.

Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
Explain why nucleophilic substitution reactions are not very common in phenols.
Assertion: Bond angle in ethers is slightly less than the tetrahedral angle.
Reason: There is a repulsion between the two bulky (–R) groups.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
