Advertisements
Advertisements
Question
Name the factors responsible for the solubility of alcohols in water.
Advertisements
Solution
Alcohols are soluble in water because of hydrogen bonding with water molecules.
\[\begin{array}{cc}
\phantom{}\ce{R - O - H ⋅⋅⋅ O - H ⋅⋅⋅ O - H ⋅⋅⋅ }\phantom{}\\
\phantom{.......}|\phantom{..........}|\phantom{.}\\
\phantom{.......}\ce{H}\phantom{.........}\ce{R}\phantom{.}
\end{array}\]
The solubility decreases with increase in size of alkyl or aryl group.
RELATED QUESTIONS
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
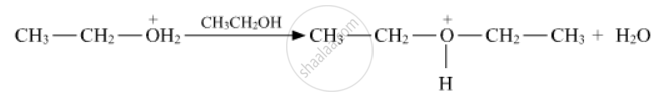
Write the mechanism of the following reaction:

Write the mechanism of the following reaction :

Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Give the equation of the following reaction:
Oxidation of propan-1-ol with alkaline KMnO4 solution.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
Write the mechanism (using curved arrow notation) of the following reaction :
Lucas reagent is ____________.
In the reduction \[\ce{R - CHO + H2 -> RCH2OH}\] the catalyst used is:
Lucas test is done to differentiate between ____________.
Which one of the following on oxidation gives a ketone?
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
\[\ce{CH3CH2OH}\] can be converted into \[\ce{CH3CHO}\] by ______.
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) |  |
| (b) |  |
| (c) |  |
Which of the following alcohols will not undergo oxidation?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
