Advertisements
Advertisements
Questions
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Explain why is ortho nitrophenol more acidic than ortho methoxyphenol?
Advertisements
Solution 1
Due to the strong –R and –I effect of the NO2 group, the electron density on the O–H bond decreases; hence, the proton is released easily.

After losing the proton, the remaining o-nitrophenoxide ion becomes stable by resonance.

Ortho-nitrophenoxide ion resonance is stable; hence, o-nitrophenol is a strong acid. On the other hand, due to the +R effect of the OCH3 group, the electron density on the O–H bond increases; hence, the removal of the proton becomes difficult.
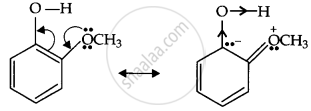
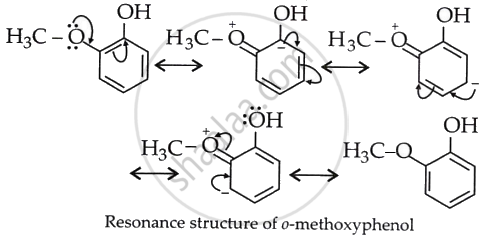
Now, the o-methoxyphenoxide ion, which remains after the loss of the proton, gets destabilised due to resonance.
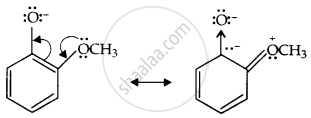
The two negative charges repel each other and destabilise the o-methoxyphenoxide ion, so o-nitrophenol is more acidic than o-methoxyphenol.
Solution 2
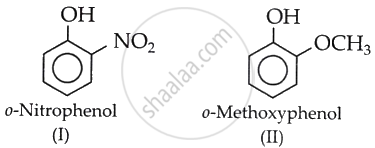
The acidity of phenol is due to the dissociation of a hydrogen ion (H+) from the hydroxyl (O–H) group. This loss increases if the O–H bond polarity intensifies and H carries a significant δ+ charge.
The presence of the strongly electron-withdrawing NO2 group at the ortho position in (I) increases the polarity of the O–H bond.
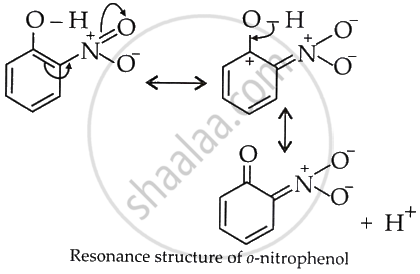
This helps in the release of H+ and increases acidity.
The reaction (II) is less acidic because –OCH3 is an electron-releasing group and increases the electron density on O of OH, and H+ release is suppressed. This can be understood from the resonance structures of II, which are:
RELATED QUESTIONS
Write the final product(s) in each of the following reactions:

Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
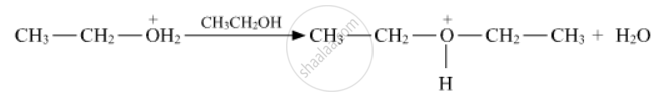
Write the mechanism (using curved arrow notation) of the following reaction :
Lucas reagent is ____________.
The compound which reacts fastest with Lucas reagent at room temperature is:
Dehydration of 2-butanol yields:
Which one of the following on oxidation gives a ketone?
Primary and secondary alcohols on the action of reduced copper give:
Alcohols react with active metals e.g. Na, K etc. to give corresponding alkoxides. Write down the decreasing order of reactivity of sodium metal towards primary, secondary and tertiary alcohols.
Explain why nucleophilic substitution reactions are not very common in phenols.
Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
The correct geometry around oxygen in CH3OCH3 is
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
