Advertisements
Advertisements
प्रश्न
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Explain why is ortho nitrophenol more acidic than ortho methoxyphenol?
Advertisements
उत्तर १
Due to the strong –R and –I effect of the NO2 group, the electron density on the O–H bond decreases; hence, the proton is released easily.

After losing the proton, the remaining o-nitrophenoxide ion becomes stable by resonance.

Ortho-nitrophenoxide ion resonance is stable; hence, o-nitrophenol is a strong acid. On the other hand, due to the +R effect of the OCH3 group, the electron density on the O–H bond increases; hence, the removal of the proton becomes difficult.
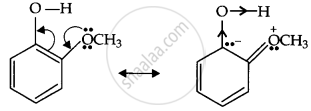
Now, the o-methoxyphenoxide ion, which remains after the loss of the proton, gets destabilised due to resonance.
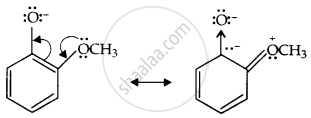
The two negative charges repel each other and destabilise the o-methoxyphenoxide ion, so o-nitrophenol is more acidic than o-methoxyphenol.
उत्तर २
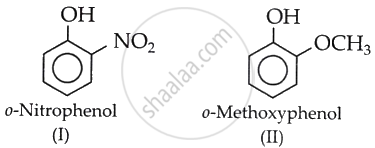
The acidity of phenol is due to the dissociation of a hydrogen ion (H+) from the hydroxyl (O–H) group. This loss increases if the O–H bond polarity intensifies and H carries a significant δ+ charge.
The presence of the strongly electron-withdrawing NO2 group at the ortho position in (I) increases the polarity of the O–H bond.
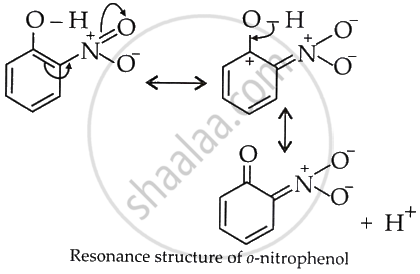
This helps in the release of H+ and increases acidity.
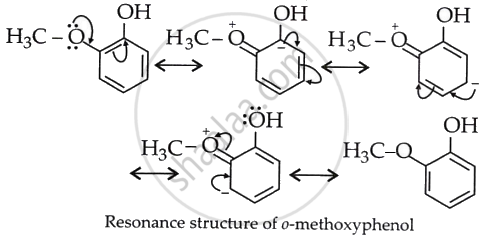
The reaction (II) is less acidic because –OCH3 is an electron-releasing group and increases the electron density on O of OH, and H+ release is suppressed. This can be understood from the resonance structures of II, which are:
संबंधित प्रश्न
Write the mechanism of the following reaction:

Give the equation of the following reaction:
Oxidation of propan-1-ol with alkaline KMnO4 solution.
Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
In the reduction \[\ce{R - CHO + H2 -> RCH2OH}\] the catalyst used is:
Which one of the following on oxidation gives a ketone?
Primary and secondary alcohols on the action of reduced copper give:
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) |  |
| (b) |  |
| (c) |  |
Name the factors responsible for the solubility of alcohols in water.
Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
Why is the C – O – H bond angle in alcohols slightly less than the tetrahedral angle whereas the C – O – C bond angle in ether is slightly greater?
The correct geometry around oxygen in CH3OCH3 is
What happens when (CH3)3 C – OH is heated with Cu/573 K?
Write the chemical equation in support of your answer.
Which of the following observation is shown by 2-phenyl ethanol with Lucas Reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene
