Advertisements
Advertisements
प्रश्न
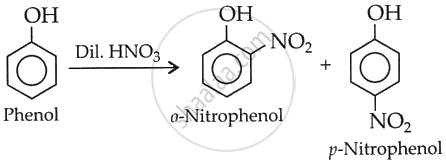
Give the equation of the following reaction:
Dilute HNO3 with phenol.
Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Write the final product(s) in each of the following reactions:

Write the mechanism of the following reaction:

Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
Lucas reagent is ____________.
In the reduction \[\ce{R - CHO + H2 -> RCH2OH}\] the catalyst used is:
By which of the following methods alcohol can be prepared in excellent yield?
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
Primary and secondary alcohols on the action of reduced copper give:
The compound which gives the most stable carbonium ion on dehydration is:
Cyclohexene is best prepared from cyclohexanol by which of the following:
\[\ce{CH3CH2OH}\] can be converted into \[\ce{CH3CHO}\] by ______.
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
Name the factors responsible for the solubility of alcohols in water.
Explain why nucleophilic substitution reactions are not very common in phenols.
Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
Which of the following observation is shown by 2-phenyl ethanol with Lucas Reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene
