Advertisements
Advertisements
प्रश्न
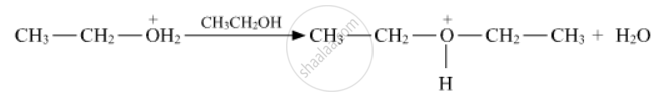
Write the mechanism (using curved arrow notation) of the following reaction :
Advertisements
उत्तर

APPEARS IN
संबंधित प्रश्न
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Write the mechanism of the following reaction:

In the reduction \[\ce{R - CHO + H2 -> RCH2OH}\] the catalyst used is:
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
Name the factors responsible for the solubility of alcohols in water.
Suggest a reagent for the following conversion.

Alcohols react with active metals e.g. Na, K etc. to give corresponding alkoxides. Write down the decreasing order of reactivity of sodium metal towards primary, secondary and tertiary alcohols.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene
