Advertisements
Advertisements
प्रश्न
Why is the C – O – H bond angle in alcohols slightly less than the tetrahedral angle whereas the C – O – C bond angle in ether is slightly greater?
Advertisements
उत्तर
The C – O – H bond angle in alcohols is slightly less than the tetrahedral angle (109.5°) because of larger repulsions between the lone pairs of electrons. For example in methanol C – O – H bond angle is 108.9°.
In ethers, the C – O – C bond angle is slightly greater than tetrahedral angle. For example in dimethyl ether C – O – C bond angle is 111.7°. The larger bond angle in ethers may be because of greater repulsions between bulkier alkyl groups as compared to one H in alcohols.
APPEARS IN
संबंधित प्रश्न
Write the mechanism of the following reaction:

Write the mechanism of the following reaction :

Give the equation of the following reaction:
Oxidation of propan-1-ol with alkaline KMnO4 solution.
Give the equation of the following reaction:
Dilute HNO3 with phenol.
Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
The compound which reacts fastest with Lucas reagent at room temperature is:
In the reduction \[\ce{R - CHO + H2 -> RCH2OH}\] the catalyst used is:
Dehydration of 2-butanol yields:
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
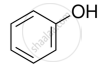
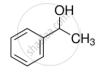
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) |  |
| (b) |  |
| (c) |  |
Name the factors responsible for the solubility of alcohols in water.
Alcohols react with active metals e.g. Na, K etc. to give corresponding alkoxides. Write down the decreasing order of reactivity of sodium metal towards primary, secondary and tertiary alcohols.
Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
Explain why nucleophilic substitution reactions are not very common in phenols.
Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
What happens when (CH3)3 C – OH is heated with Cu/573 K?
Write the chemical equation in support of your answer.
Which of the following observation is shown by 2-phenyl ethanol with Lucas Reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
