Advertisements
Advertisements
प्रश्न
Give the equation of the following reaction:
Oxidation of propan-1-ol with alkaline KMnO4 solution.
Advertisements
उत्तर
\[\ce{\underset{propan-1-ol}{CH3CH2CH2OH} + 2[O] ->[Alkaline KMnO4][Oxidation] \underset{Propanoic acid}{CH3CH2COOH} + H2O}\]
APPEARS IN
संबंधित प्रश्न
Write the mechanism of the following reaction :

Show how will you synthesize pentan-1-ol using a suitable alkyl halide.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
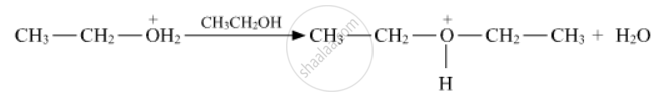
Write the mechanism (using curved arrow notation) of the following reaction :
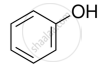
Lucas test is used for the detection of _____________.
Dehydration of 2-butanol yields:
Lucas test is done to differentiate between ____________.
Which one of the following on oxidation gives a ketone?
Cyclohexene is best prepared from cyclohexanol by which of the following:
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
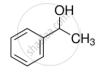
The process of converting alkyl halides into alcohols involves ______.
Explain why nucleophilic substitution reactions are not very common in phenols.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
