Advertisements
Advertisements
प्रश्न
Explain how does the −OH group attached to a carbon of benzene ring activate it towards electrophilic substitution?
Advertisements
उत्तर १
Phenol is considered to be a resonance hybrid of the following structures:

The +R effect of the –OH group increases the electron density on the benzene ring, which makes the attack of the electrophile easier. Hence, the presence of the –OH group activates the benzene ring towards electrophilic substitution reactions. Since the electron density at the ortho and para positions is relatively high, electrophilic substitution is mainly more at the ortho and para positions.
उत्तर २
In an electrophilic substitution reaction, an electron-deficient species attacks the benzene ring, which is electron-rich. When an –OH group is attached to the benzene ring, it increases the ring's electron density, making it more reactive and a good site for electrophilic attack.
The increase in electron density can be visualised as:
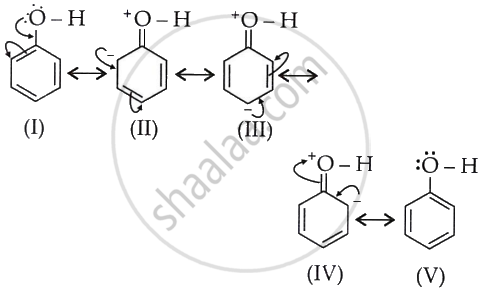
Structures (I)-(V) indicate that the addition of a hydroxyl group to benzene has increased the electron density (negative charge) on the carbon atoms of the ring, particularly at C-2, C-4, and C-6. Therefore, it is said that the ring has been activated towards electrophiles, which are drawn to the increased electron density.
APPEARS IN
संबंधित प्रश्न
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
Butan-1-ol
Intermolecular hydrogen bonding is strongest in ______.
Phenols do not react with one of the following:
Acidity of phenol is due to ____________.
The ionization constant of phenol is higher than that of ethanol because ____________.
In the following compounds:
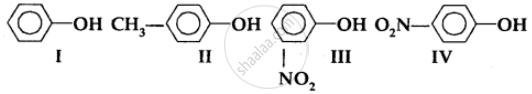
The order of acidity is
In CH3CH2OH, the bond that undergoes heterolytical change most readily is ____________.
Strength of acidity is in order:

Arrange the following in decreasing order of acidic character:

Out of o-nitrophenol and o-cresol which is more acidic?
For the pair phenol and cyclohexanol, answer the following:
Why is phenol more acidic than cyclohexanol?
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
2-Methylbutan-2-ol
Give the structure of the product you would expect when the following alcohol reacts with HBr.
Butan-1-ol
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
Butan-1-ol
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
2-Methylbutan-2-ol
Compare acidity of phenol with that of ethanol.
