Advertisements
Advertisements
प्रश्न
Arrange the following in the increasing order of their property indicated:
4-Nitrobenzoic acid, benzoic acid, 3,4-Dinitrobenzoic acid, 4-Methoxy benzoic acid (Acid strength)
Advertisements
उत्तर
The increasing order of acidic strength of the following compounds is 4-methoxy benzoic acid < benzoic acid < 4-nitrobenzoic acid < 3,4-dinitrobenzoic acid. (Due to the presence of electron withdrawing group).
APPEARS IN
संबंधित प्रश्न
Account for the following:
o-nitrophenol is more steam volatile than p-nitrophenol.
Phenols do not react with one of the following:
Phenol is more acidic than alcohol because ____________.
Which one of the following compounds has the most acid nature?
Arrange the following in decreasing order of acidic character:

Mark the correct order of decreasing acid strength of the following compounds.
| (a) | 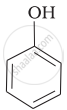 |
| (b) | 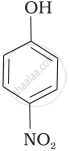 |
| (c) | 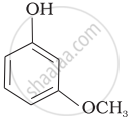 |
| (d) | 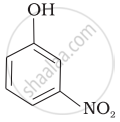 |
| (e) | 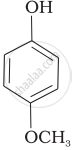 |
In the following compounds:
 |
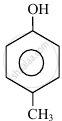 |
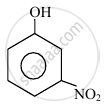 |
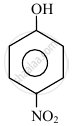 |
| (I) | (II) | (III) | (IV) |
The order to acidity is ______.
Which one of the following has the lowest pKa value?
For the pair phenol and cyclohexanol, answer the following:
Why is phenol more acidic than cyclohexanol?
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
Butan-1-ol
