Advertisements
Advertisements
प्रश्न
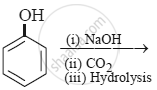
Give two reactions that show the acidic nature of phenol.
Advertisements
उत्तर
- Reaction with sodium: Phenol reacts with active metals such as sodium to liberate hydrogen.

- Reaction with NaOH: Phenol dissolves in NaOH to form sodium phenoxide and water.

APPEARS IN
संबंधित प्रश्न
Explain how does the −OH group attached to a carbon of benzene ring activate it towards electrophilic substitution?
Account for the following:
o-nitrophenol is more steam volatile than p-nitrophenol.
The product obtained from the reaction is:
Phenols do not react with one of the following:
Phenol is more acidic than alcohol because ____________.
In CH3CH2OH, the bond that undergoes heterolytical change most readily is ____________.
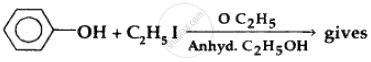
Strength of acidity is in order:

Which of the following compounds is most acidic?
Arrange the following in decreasing order of acidic character:

Assertion: o-Nitrophenol is less soluble in water than the m- and p-isomers.
Reason: m- and p- Nitrophenols exist as associated molecules.
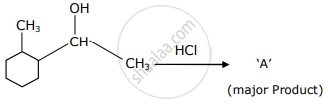
Which is the final product ‘A’ (major) in the given reaction?
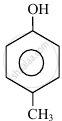
In the following compounds:
 |
 |
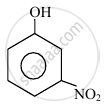 |
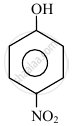 |
| (I) | (II) | (III) | (IV) |
The order to acidity is ______.
Which one of the following has the lowest pKa value?
Give the structure of the product you would expect when the following alcohol reacts with HBr.
Butan-1-ol
