Advertisements
Advertisements
प्रश्न
Give two reactions that show the acidic nature of phenol.
Advertisements
उत्तर
- Reaction with sodium: Phenol reacts with active metals such as sodium to liberate hydrogen.

- Reaction with NaOH: Phenol dissolves in NaOH to form sodium phenoxide and water.

APPEARS IN
संबंधित प्रश्न
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
Butan-1-ol
Explain how does the −OH group attached to a carbon of benzene ring activate it towards electrophilic substitution?
Account for the following:
o-nitrophenol is more steam volatile than p-nitrophenol.
Phenols do not react with one of the following:
Phenol is more acidic than alcohol because ____________.
Acidity of phenol is due to ____________.
The ionization constant of phenol is higher than that of ethanol because ____________.
In CH3CH2OH, the bond that undergoes heterolytical change most readily is ____________.
Phenol reacts with Br2 in CS2 at low temperature to give ____________.
Strength of acidity is in order:

Arrange the following in decreasing order of acidic character:

Which of the following statements is true:
Out of o-nitrophenol and o-cresol which is more acidic?
Assertion: o-Nitrophenol is less soluble in water than the m- and p-isomers.
Reason: m- and p- Nitrophenols exist as associated molecules.
In the following compounds:
 |
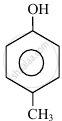 |
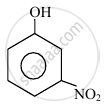 |
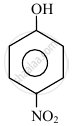 |
| (I) | (II) | (III) | (IV) |
The order to acidity is ______.
For the pair phenol and cyclohexanol, answer the following:
Why is phenol more acidic than cyclohexanol?
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
Butan-1-ol
Compare acidity of phenol with that of ethanol.
