Advertisements
Advertisements
प्रश्न
Which one of the following has the lowest pKa value?
पर्याय
CH3 – COOH
O2N–CH2–COOH
Cl–CH2–COOH
HCOOH
Advertisements
उत्तर
O2N–CH2–COOH
Explanation:
It is simpler to remove an electron since the –NO2 group is an electron-withdrawing group. As a result, the acidity is stronger, and the pKa value is lower.
APPEARS IN
संबंधित प्रश्न
Write the equation involved in the acetylation of Salicylic acid.
Give two reactions that show the acidic nature of phenol.
Phenols do not react with one of the following:
Phenol reacts with Br2 in CS2 at low temperature to give ____________.
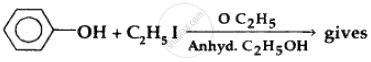
Strength of acidity is in order:

Which of the following compounds is most acidic?
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
Butan-1-ol
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
2-Methylbutan-2-ol
Compare acidity of phenol with that of ethanol.
