Advertisements
Advertisements
प्रश्न
Compare acidity of phenol with that of ethanol.
Advertisements
उत्तर १
Phenol is more acidic than ethanol. This is because the phenoxide ion obtained after the release of a proton from phenol becomes stable through resonance, whereas the ethoxide ion (after the release of a proton from ethanol) is not stable.
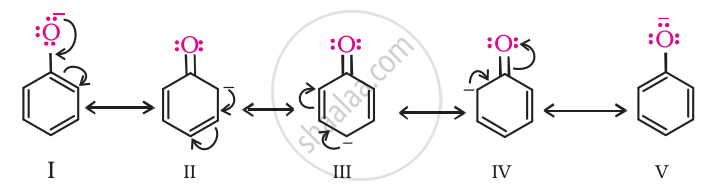
उत्तर २
Compared with ethanol, phenols are more acidic. They lose their H atom as H+ more readily than alcohols. The behaviour of phenol can be attributed to resonance stabilisation arising from the contributing structures of the corresponding phenoxide ion.
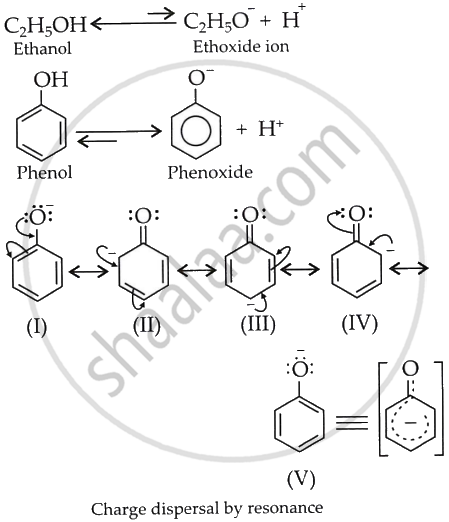
The resonance stabilisation from the contributing structures (I)-(V) sufficiently offsets the O–H bond dissociation energy, rendering phenol acidic.
No resonance structures exist for the ethoxide ion; hence, the conversion of ethanol to ethoxide is not encouraged under standard conditions. Therefore, ethanol exhibits lower acidity compared to phenol.
APPEARS IN
संबंधित प्रश्न
Give two reactions that show the acidic nature of phenol.
Account for the following:
o-nitrophenol is more steam volatile than p-nitrophenol.
The product obtained from the reaction is:
Intermolecular hydrogen bonding is strongest in ______.
Phenols do not react with one of the following:
In CH3CH2OH, the bond that undergoes heterolytical change most readily is ____________.
Strength of acidity is in order:

Which of the following statements is true:
Assertion: o-Nitrophenol is less soluble in water than the m- and p-isomers.
Reason: m- and p- Nitrophenols exist as associated molecules.
Phenol is used in the manufacture of
Arrange the following in the increasing order of their property indicated:
4-Nitrobenzoic acid, benzoic acid, 3,4-Dinitrobenzoic acid, 4-Methoxy benzoic acid (Acid strength)
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
2-Methylbutan-2-ol
Give the structure of the product you would expect when the following alcohol reacts with HBr.
Butan-1-ol
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
Butan-1-ol
