Advertisements
Advertisements
Question
Which one of the following has the lowest pKa value?
Options
CH3 – COOH
O2N–CH2–COOH
Cl–CH2–COOH
HCOOH
Advertisements
Solution
O2N–CH2–COOH
Explanation:
It is simpler to remove an electron since the –NO2 group is an electron-withdrawing group. As a result, the acidity is stronger, and the pKa value is lower.
APPEARS IN
RELATED QUESTIONS
Explain how does the −OH group attached to a carbon of benzene ring activate it towards electrophilic substitution?
Account for the following:
o-nitrophenol is more steam volatile than p-nitrophenol.
The product obtained from the reaction is:
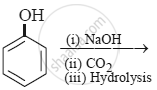
Phenol is more acidic than alcohol because ____________.
The ionization constant of phenol is higher than that of ethanol because ____________.
Which of the following compounds is most acidic?
What is the correct order of reactivity of alcohols in the following reaction?
\[\ce{R-OH + HCl ->[ZnCl2] R-Cl + H2O}\]
Which is the final product ‘A’ (major) in the given reaction?
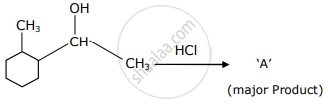
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
2-Methylbutan-2-ol
Give the structure of the product you would expect when the following alcohol reacts with HBr.
Butan-1-ol
