Advertisements
Advertisements
Question
What is the correct order of reactivity of alcohols in the following reaction?
\[\ce{R-OH + HCl ->[ZnCl2] R-Cl + H2O}\]
Options
1° > 2° > 3°
1° < 2° > 3°
3° > 2° > 1°
3° > 1° > 2°
Advertisements
Solution
3° > 2° > 1°
Explanation:
The given reaction is nucleophilic substitution reaction in which –OH group is replaced by –Cl. Tertiary alcohols, when react with \[\ce{HCl}\] in presence of \[\ce{ZnCl2}\], form tertiary carbocation.
This intermediate 3° carbocation is more stable than 2° carbocation as well as 1° carbocation. The higher the stability of intermediate, the higher will be the reactivity of reactant molecule.
So, the order of reactivity of alcohols in the given reaction is 3° > 2° > 1°.
APPEARS IN
RELATED QUESTIONS
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
Butan-1-ol
Explain how does the −OH group attached to a carbon of benzene ring activate it towards electrophilic substitution?
Account for the following:
o-nitrophenol is more steam volatile than p-nitrophenol.
The product obtained from the reaction is:
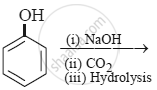
Phenol is more acidic than alcohol because ____________.
Acidity of phenol is due to ____________.
The ionization constant of phenol is higher than that of ethanol because ____________.
In the following compounds:
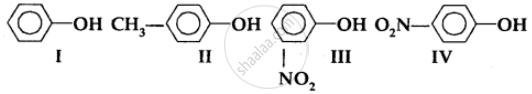
The order of acidity is
Phenol reacts with Br2 in CS2 at low temperature to give ____________.
Which one of the following compounds has the most acid nature?
Mark the correct order of decreasing acid strength of the following compounds.
| (a) | 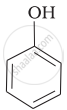 |
| (b) | 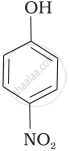 |
| (c) | 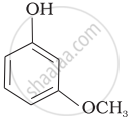 |
| (d) | 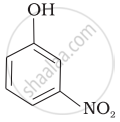 |
| (e) | 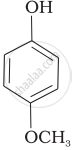 |
Arrange the following in the increasing order of their property indicated:
4-Nitrobenzoic acid, benzoic acid, 3,4-Dinitrobenzoic acid, 4-Methoxy benzoic acid (Acid strength)
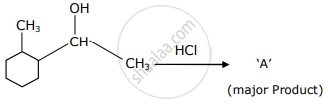
Which is the final product ‘A’ (major) in the given reaction?
In the following compounds:
 |
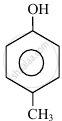 |
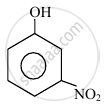 |
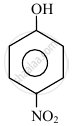 |
| (I) | (II) | (III) | (IV) |
The order to acidity is ______.
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
2-Methylbutan-2-ol
