Advertisements
Advertisements
प्रश्न
Give the structure of the product you would expect when the following alcohol reacts with HBr.
2-Methylbutan-2-ol
Advertisements
उत्तर
\[\begin{array}{cc}
\ce{CH3}\phantom{..........................................}\ce{CH3}\phantom{..............}\\
|\phantom{................................................}|\phantom{..................}\\
\ce{CH3 - C - CH2CH3 + HBr ->[\Delta] CH3 - C - CH2CH3 + H2O}\\
|\phantom{................................................}|\phantom{..................}\\
\phantom{.}\ce{OH}\phantom{................................}\ce{\underset{2-bromo-2-methylbutane}{Br}}\phantom{......}\\
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
Butan-1-ol
Phenol is more acidic than alcohol because ____________.
Acidity of phenol is due to ____________.
In the following compounds:
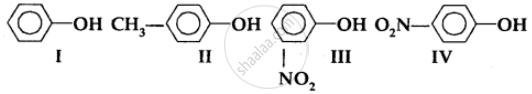
The order of acidity is
In CH3CH2OH, the bond that undergoes heterolytical change most readily is ____________.
Phenol reacts with Br2 in CS2 at low temperature to give ____________.
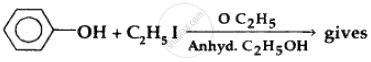
Which of the following compounds is most acidic?
Which one of the following compounds has the most acid nature?
Arrange the following in decreasing order of acidic character:

Assertion: o-Nitrophenol is less soluble in water than the m- and p-isomers.
Reason: m- and p- Nitrophenols exist as associated molecules.
Phenol is used in the manufacture of
Arrange the following in the increasing order of their property indicated:
4-Nitrobenzoic acid, benzoic acid, 3,4-Dinitrobenzoic acid, 4-Methoxy benzoic acid (Acid strength)
In the following compounds:
 |
 |
 |
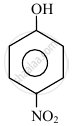 |
| (I) | (II) | (III) | (IV) |
The order to acidity is ______.
Which one of the following has the lowest pKa value?
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
Butan-1-ol
Compare acidity of phenol with that of ethanol.
