Advertisements
Advertisements
प्रश्न
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
Butan-1-ol
Advertisements
उत्तर
\[\ce{CH3CH2CH2CH2OH + HCl_{(conc.)}->[ZnCl2]No reaction at room temperature}\]
APPEARS IN
संबंधित प्रश्न
Write the equation involved in the acetylation of Salicylic acid.
Give two reactions that show the acidic nature of phenol.
Explain how does the −OH group attached to a carbon of benzene ring activate it towards electrophilic substitution?
Account for the following:
o-nitrophenol is more steam volatile than p-nitrophenol.
Intermolecular hydrogen bonding is strongest in ______.
Phenol is more acidic than alcohol because ____________.
The ionization constant of phenol is higher than that of ethanol because ____________.
In the following compounds:
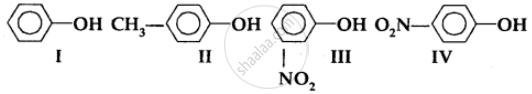
The order of acidity is
In CH3CH2OH, the bond that undergoes heterolytical change most readily is ____________.
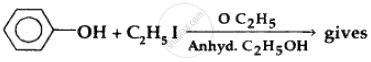
Which of the following compounds is most acidic?
Which of the following statements is true:
Assertion: o-Nitrophenol is less soluble in water than the m- and p-isomers.
Reason: m- and p- Nitrophenols exist as associated molecules.
Phenol is used in the manufacture of
Arrange the following in the increasing order of their property indicated:
4-Nitrobenzoic acid, benzoic acid, 3,4-Dinitrobenzoic acid, 4-Methoxy benzoic acid (Acid strength)
For the pair phenol and cyclohexanol, answer the following:
Why is phenol more acidic than cyclohexanol?
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
2-Methylbutan-2-ol
