Advertisements
Advertisements
प्रश्न
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
Butan-1-ol
Advertisements
उत्तर
\[\ce{\underset{Butan-1-ol}{CH3CH2CH2CH2OH} + SOCl2 ->[\Delta]\underset{1-chlorobutane}{CH3CH2CH2CH2Cl} + SO2 + HCl}\]
APPEARS IN
संबंधित प्रश्न
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
Butan-1-ol
The product obtained from the reaction is:
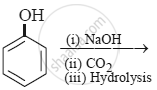
The ionization constant of phenol is higher than that of ethanol because ____________.
Strength of acidity is in order:

Which one of the following compounds has the most acid nature?
Arrange the following in decreasing order of acidic character:

Which of the following statements is true:
Mark the correct order of decreasing acid strength of the following compounds.
| (a) | 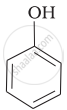 |
| (b) | 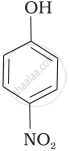 |
| (c) | 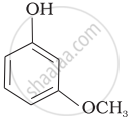 |
| (d) | 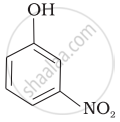 |
| (e) | 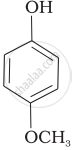 |
Out of o-nitrophenol and o-cresol which is more acidic?
Phenol is used in the manufacture of
Arrange the following in the increasing order of their property indicated:
4-Nitrobenzoic acid, benzoic acid, 3,4-Dinitrobenzoic acid, 4-Methoxy benzoic acid (Acid strength)
Which is the final product ‘A’ (major) in the given reaction?
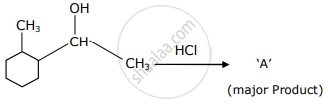
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
2-Methylbutan-2-ol
Give the structure of the product you would expect when the following alcohol reacts with HBr.
2-Methylbutan-2-ol
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
2-Methylbutan-2-ol
Compare acidity of phenol with that of ethanol.
