Advertisements
Advertisements
Question
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
Butan-1-ol
Advertisements
Solution
\[\ce{\underset{Butan-1-ol}{CH3CH2CH2CH2OH} + SOCl2 ->[\Delta]\underset{1-chlorobutane}{CH3CH2CH2CH2Cl} + SO2 + HCl}\]
APPEARS IN
RELATED QUESTIONS
Give two reactions that show the acidic nature of phenol.
Account for the following:
o-nitrophenol is more steam volatile than p-nitrophenol.
The product obtained from the reaction is:
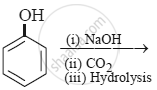
Phenols do not react with one of the following:
Phenol is more acidic than alcohol because ____________.
Acidity of phenol is due to ____________.
The ionization constant of phenol is higher than that of ethanol because ____________.
In the following compounds:
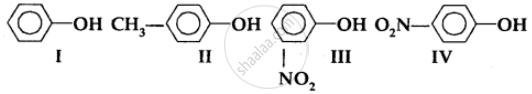
The order of acidity is
Phenol reacts with Br2 in CS2 at low temperature to give ____________.
Which one of the following compounds has the most acid nature?
Arrange the following in decreasing order of acidic character:

What is the correct order of reactivity of alcohols in the following reaction?
\[\ce{R-OH + HCl ->[ZnCl2] R-Cl + H2O}\]
Out of o-nitrophenol and o-cresol which is more acidic?
Assertion: o-Nitrophenol is less soluble in water than the m- and p-isomers.
Reason: m- and p- Nitrophenols exist as associated molecules.
For the pair phenol and cyclohexanol, answer the following:
Why is phenol more acidic than cyclohexanol?
Give the structure of the product you would expect when the following alcohol reacts with HBr.
2-Methylbutan-2-ol
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
2-Methylbutan-2-ol
